Sugar beet seedling rust is a heteroecious rust disease caused by the macrocyclic pathogen Puccinia subnitens Dietel (syn. P. aristidae Tracy). Only the pycnial and aecial stages occur on sugar beet, while the other stages (uredial and telial) infect various species of saltgrass (Distichlis spp.) and needlegrass (Aristida spp.) (12,19,20). The primary documented host for P. subnitens is the inland saltgrass (Distichlis spicata L. Greene) (7,11). Inland saltgrass is a warm-season grass native to arid areas of the western United States, and is also commonly found in brackish, marshy areas or highly saline soils (11). The plant is very drought tolerant and thrives in the strongly alkaline soils throughout the North American High Plains and West.
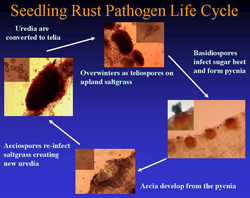
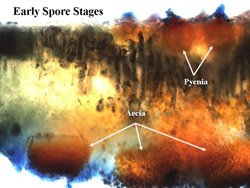
In early spring, teliospores germinate from telial pustules after overwintering on inland saltgrass to produce basidiospores, which then are blown by wind where they can infect sugar beets or a number of other alternate hosts (see below). The basidiospore infections give rise sequentially to the pycnial (also referred to as spermagonial) and aecial spore stages (Fig. 1). Pycnial lesions are circular and light yellow, measuring 2 to 5 mm in diameter (Fig. 2A). Flask-shaped pycnia containing receptive hyphae and spermatia (Fig. 3) are found embedded within lesions and are located primarily on the upper leaf surface (Fig. 4). Aecia, which develop directly from the pycnia after fertilization, are usually found on the lower leaf surface directly below the pycnia (Fig. 4). The aecia consist of clusters of yellowish-orange, rounded structures (Figs. 2B and 5) that become cup-shaped after rupturing (Fig. 6), releasing spores, and are often found aggregated in circular patterns (Figs. 2B and 7). Newly formed aeciospores then re-infect the inland saltgrass, creating new uredia and telia, completing the life cycle (Fig. 1).
 |
|
 |
|
Fig. 1. Life cycle of the sugar beet seedling rust pathogen (Puccinia subnitens). |
|
Fig. 2. Circular, light-yellow pycnial lesions on the upper surface of a sugar beet cotyledon (A); yellowish-orange aecial pustules arranged in rings on the lower cotyledon surface (B). |
 |
|
 |
|
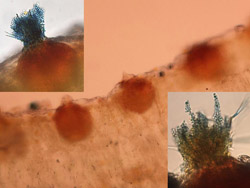
Fig. 3. Thin section of seedling rust-infected sugar beet leaf containing pycnia embedded in the epidermal surface. Close-up of one pycnium exhibiting receptive hyphae and spermatia (insets). |
|
Fig. 4. Thin section of infected sugar beet leaf showing flask-shaped pycnia on the top leaf surface, and aecial cups and developing aeciospores on the bottom leaf surface. |
 |
|
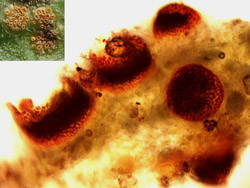 |
|
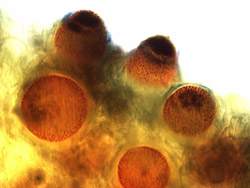
Fig. 5. Thin section of an aecial lesion from an infected sugar beet leaf demonstrating the immature aecia prior to spore release. |
|
Fig. 6. Thin section of an aecial lesion from an infected sugar beet leaf showing the formation of the cup-shaped aecia after rupturing with mature empty aecia after spore release (inset). |
| |
 |
|
| |
Fig. 7. Young aecia arranged in circular patterns on the underside of a common lambsquarters leaf. |
|
The first published description of Puccinia subnitens has been attributed to Paul Dietel in 1895 (6). Afterwards, this pathogen was commonly recognized on various Distichlis species from coast to coast across North America, and was particularly prevalent throughout the western United States. A close association between P. subnitens infections on saltgrass and aecial rust infections on common lambsquarters (Chenopodium album) (Fig. 8) very similar to that on sugar beet was also widely observed across the Great Plains and Western United States from North Dakota and Montana to New Mexico and Arizona to California (5).
| |
 |
|
| |
Fig. 8. Distribution on lambsquarters of pycnia (A) and aecia (B) on the upper and lower leaf surfaces, respectively. |
|
Prior to 1904, C. album was the only known aecial host for this rust (P. subnitens). However, in the summer of 1904, the Reverend John Mallory Bates postulated that aecia he observed in Nebraska infecting species of Cleome (Beeweed), Sophia (tansymustard), Lepidium (pepperweed), Erysimum (wallflower), and Salsola (Russian thistle) were related to P. subnitens (1,4). J. M. Bates was an Episcopalian clergyman widely known and respected as an amateur naturalist and botanist. Although these studies were only a hobby for him, he was still recognized as an authority on plant rust diseases and ornithology.
Bates communicated his observations to the noted rust pathologist, J. C. Arthur and sent him teliospores from Distichlis, and infected seedlings of Cleome serrulata and C. album covered profusely with the yellowish-orange-colored aecia. As the season progressed, Bates also sent Arthur freshly collected specimens containing aecia on Roripa sinulata (spreading yellowcress), Sophia incisa, and Salsola tragus, all of which he thought belonged to the same rust species. Arthur pronounced the aecia from these plants to be identical to P. subnitens and confirmed Bates’ deductions later that summer by inoculating these same host plants with teleospores and observing the formation of new aecia (1). Arthur considered it exceptional that P. subnitens was capable of infecting this number and diversity of host plants, a result which became a watershed concept for rust investigations at the time (1,2).
Ellsworth Bethel, with the Colorado State Museum in Denver, also found the same fungus distributed throughout Colorado from many of the same plant species as that of Bates in Nebraska. By 1934, the combined works of Bethel, Arthur, and others further confirmed and expanded the host range of P. subnitens to include a remarkable 84 species in 19 families and 52 genera (2,4,5).
Although it has been readily demonstrated that the aecial form of P. subnitens is often found on many disparate weed species, infection on cultivated, economic host plants is restricted to members of the Chenopodiaceae, including beet, mangel, Swiss chard, and spinach (1,18,19). On spinach, the disease is called "red rust" to differentiate it from the chronic spinach disease "white rust," which is not a rust pathogen at all, but a water mold (oomycete) in the genus Albugo (17). Symptoms of red rust on spinach include small yellow spots with red centers (pycnial pustules) that occur on upper leaf surfaces (Fig. 9). Aecial clusters can then be found scattered profusely on both upper and lower leaf surfaces (Fig. 10), often arranged in rings (Fig. 10, inset) in a similar manner as that of the previously discussed sugar beets and lambsquarters species.
 |
|
 |
|
Fig. 9. Faint pycnial lesions of P. subnitens scattered across the upper surface of spinach leaf (courtesy of H. F. Schwartz). |
|
Fig. 10. Conspicuous aecial lesions of P. subnitens on both upper and lower surfaces of spinach leaves with a close-up (inset) of aecial lesions showing arrangement of pustules on spinach leaf (both images courtesy of H. F. Schwartz). |
The first apparent report implicating this pathogen with serious crop damage was from the Pacific Northwest in 1922 (3). H. P. Barss reported that vegetable growers in the Walla Walla Valley of Oregon and Washington sustained severe losses to their spinach crops during the spring of 1922. He also mentioned that this condition had never been observed in recent memory by anyone associated with spinach production in this region, making it a new report (3). Because no other rust disease of spinach was known at that time, it was postulated that some indigenous rust pathogen was responsible for the infection. P. subnitens was suspected as the cause (implying that it was familiar to Barss) and after a brief search for the saltgrass, it was found to be one of the three most commonly found grass species growing in the vicinity of spinach production fields in this area.
It was not until the 1940s, that this disease was heard from again. It was first identified on spinach from Colorado from multiple fields over two sequential seasons (1943-1944) (14,15). Since that time the disease has appeared sporadically and severe losses in Colorado spinach production have been attributed to the same pathogen in 1952 and 1959 with additional reports of outbreaks in 1996 and 1999 (17).
A survey conducted in the summer of 1959 identified the telial stage of the pathogen infecting saltgrass from 14 distinct counties, widely scattered across Colorado (12). Thus it was concluded that spores from the saltgrass must have been the sources of infection for spinach during 1943-1944 and the epidemics of the 1950s. It was additionally noted that cultivated plants that were hosts for the aecial stage (spinach and sugar beets) were not always present in the vicinity of the infected saltgrass to complete the life cycle, but was concluded that wild plants in a variety of families were available and must have served as aecial hosts (12).
The damage to Colorado spinach production was induced both in the field and in storage while being transported to market. The first epidemics in 1943-1944 reported that infections within some fields were minor while other fields had to be destroyed due to severe leaf spotting on 50 to 90% of plants, making the plants unmarketable (14,15). In the 1950s the disease was shown to progress rapidly on plants over several days while being shipped to market, rendering them unsalable upon arrival at their terminal destination (16). This clearly illustrated one of the distinct characteristics of the disease on spinach. Spinach plants obviously became infected in the field, but decomposed rapidly after infections were undetected by packers prior to shipping. Even with our advanced shipping technology today, this disease is still difficult for packers to detect, and poses a problem because disease can develop so rapidly during transit (16,17).
The first published report of this disease on sugar beets was also from Colorado, by Pool and McKay in 1914 (13). They found the rust occurring frequently during the 1912-1913 seasons from multiple fields in the Arkansas Valley near Rocky Ford, CO. During May and June, the pathogen produced aecial lesions primarily on young sugar beet cotyledons, but also caused some moderate damage on infected swollen crowns. They also noted lambsquarters that were heavily infected with aecial lesions within the same locations. By early July the disease intensity had diminished and was no longer an issue. They further added that the telial form was found abundantly on saltgrass stands near fields during the sugar beet epidemics, followed by new uredia being identified in early June before being converted back to telia in late July on saltgrass (13).
Pool and McKay (14) additionally mentioned that conversations with agronomists ascertained that this disease had been seen only once before in one location, while correspondence with J. C. Arthur revealed that Arthur had found the pathogen once before on sugar beets in Colorado, but apparently had not yet published on it. They finally concluded that the disease was not severe during these two seasons, but that if conditions were conducive, significant damage could be realized to young plants due to the profusion of infected saltgrass stands within close proximity of sugar beet fields (13).
In mid May 2009, young sugar beet plants were first observed exhibiting signs indicative of sugar beet seedling rust in a field near Bayard, NE (Fig. 11), after an extended period of unusually cool and wet weather had persisted throughout the western Nebraska Panhandle in April and early May (8). Temperatures during these two months were found to be 13°C cooler with rainfall 12 cm higher than the 30-year average for this area. Disease incidence in this beet field approached 25%, although lesions were restricted primarily to the cotyledons.
| |
 |
|
| |
Fig. 11. Field-infected, young sugar beet plants with pycnial lesions on cotyledons. |
|
This particular field was then monitored 5 to 6 times during the remainder of the entire season to determine whether the infection could also be due to beet rust, a distinct disease caused by the macrocyclic, autoecious pathogen Uromyces betae (8). Since no uredial pustules were ever observed on sugar beet plants, beet rust was effectively ruled out as the causative factor in this field. Pycnial lesions were circular, and yellowish, 2-5 mm in diameter (Fig. 2A) embedded with flask-shaped pycnia (Fig. 3), with globoid, verrucose aeciospores measuring approximately 17-22 µm × 15-20 µm, characteristic of P. subnitens (7). It was also additionally noted that this particular infested sugar beet field was surrounded by stands of inland saltgrass also infected with the telial stage of a rust pathogen (Fig. 12), presumed to be P. subnitens, but not confirmed, which provided further evidence hinting at the pathogen’s identity (8).
| |
 |
|
| |
Fig. 12. Upland saltgrass infected with the telial stage of P. subnitens. |
|
Based on this initial identification of the pathogen in Morrill Co., Nebraska in mid May 2009, a survey of sugar beet production fields in western Nebraska was conducted between late May and mid June to further document the incidence and number of fields infested with seedling rust caused by P. subnitens. After similar environmental conditions were experienced in the late spring of 2010, another similar survey was conducted in May and June 2010 from more than thirty locations throughout western Nebraska (8,10). In fact, the environment was so conducive for rust development over these two years that a similar phenomenon was observed with sunflower rust and the widespread occurrence of the early spore stages on wild sunflowers and volunteers across the region (9).
Over this two-year period, 88 locations from eight western Nebraska counties (Scotts Bluff, Morrill, Box Butte, Banner, Kimball, Sioux, Cheyenne, and Sheridan) were scouted and 47 were identified with plants infected with pycnia and/or aecia of P. subnitens. Forty-one of these locations were sugar beet fields, with 36% (15/41) of the fields additionally containing infected lambsquarters (10). Infected lambsquarters were identified from several other locations throughout the area, including ditchbanks, an uncultivated field, a home garden, and a flower bed in a restaurant parking lot in Scottsbluff, NE.
For both years, the majority of the pathogen-infested fields (85%) were located within the North Platte Valley (Scotts Bluff and Morrill counties) (10). Although the pathogen was readily found throughout both years, the incidence within and among sites was lower in 2010 with only 30% of the monitored locations yielding infected plants compared to 65% in 2009 (8,10).
Sugar beet seedling rust, caused by Puccinia subnitens, is a disease that has rarely been observed in sugar beet production. In fact, to my knowledge the two reports from Nebraska in 2010-2011 (8,10) were the first published records of the natural occurrence in the field of this disease on sugar beets since the original communication in 1914 by Pool and McKay (13).
The initial report of this disease indicated that infection was restricted to cotyledons (Fig. 13) with symptoms being found occasionally on the first true leaves (Fig. 14), hence the name seedling disease (7,13). This two year survey from Nebraska further documented the manifestation of multiple infections from many of the surveyed sites, with 20% of fields (18 of 88) from both years yielding plants with pycnial lesions on newly emerged leaves in mid to late June (10). These observations indicate the release of multiple flushes of basidiospores of the pathogen after May from the alternate host, followed by new infections on sugar beets long past the cotyledon or first true leaf stage (up to the 5-6 true leaf stage) (Fig. 15).
 |
|
 |
|
Fig. 13. Field-infected, young sugar beet plant exhibiting both pycnial and aecial lesions, but restricted to the cotyledons. |
|
Fig. 14. Pycnial lesion on one of the first true leaves of an infected sugar beet plant. |
| |
 |
|
| |
Fig. 15. Sugar beet plant in the 5-6 leaf growth stage exhibiting a pycnial lesion of P. subnitens. Close-up of pycnial lesion (inset). |
|
The Reverend J. M. Bates cleverly observed infection on numerous weed species in Nebraska and correlated these infections with the disease of saltgrass. However, the discovery in 2009 represented the first report of the disease on sugar beets in Nebraska and the first in almost 100 years from a naturally-occurring field epidemic (8,10). Not only has this investigation been valuable by documenting a new report for a rarely occurring disease, but it has also provided a foundation for recognizing future outbreaks. By correctly diagnosing the disease rapidly, fungicide applications were avoided, which would otherwise have been made due to the unknown nature of the problem.
Regardless of the recent reports from Nebraska (8,10), seedling rust infection of sugar beets is still very rare. It was noted readily in 2009 and less extensively in 2010, but not at all in 2011, although it was commonly found on lambsquarters in all three years. This indicates that the primary host, saltgrass was present somewhere in the immediate vicinity of affected fields. These observations further suggest that environmental conditions for disease were still conducive, but perhaps also intimated that lambsquarters is more susceptible to infection than sugar beets. Sugar beet seedling rust, in general, is thought to be favored by wet and cool conditions in the spring, but optimal conditions for disease development have not been studied extensively in any host due to the novelty of its existence, even after more than 100 years of sugar beet production in this region.
All images courtesy of R. M. Harveson except for Figures 9 and 10, which are courtesy of Howard F. Schwartz, Colorado State University.
1. Arthur, J. C. 1905. Cultures of the Uredineae in 1904. J. Mycol. 11:50-67.
2. Arthur, J. C. 1934. Manual of the Rusts of the United States and Canada. Purdue Res. Found., Lafayette, IN.
3. Barss, H. P. 1922. Destructive rust (Puccinia subnitens Dietel.) on spinach in the northwest. Phytopathology 12:446.
4. Bethel, E. 1917. Puccinia subnitens and its aecial hosts. Phytopathology 7:92-94.
5. Bethel, E. 1919. Puccinia subnitens and its aecial hosts II. Phytopathology 9:193-201.
6. Dietel, P. 1895. New North American Uredineae. Erythea 3: 81:57-82.
7. Hanson, L. E. 2009. Beet rust and seedling rust. Pages 12-13 in: Compendium of Beet Diseases and Pests, 2nd Edn. R. M. Harveson, L. E. Hanson, and G. L. Hein, eds. American Phytopathological Society, St. Paul, MN.
8. Harveson, R. M. 2010. First report of sugar beet seedling rust caused by Puccinia subnitens in Nebraska. Online. Plant Health Progress doi:10.1094/PHP-2010-0315-03-BR.
9. Harveson, R. M. 2010. First report of the early spore stages of sunflower rust, caused by Puccinia helianthi, in Nebraska. Online. Plant Health Progress doi:10.1094/PHP-2010-0315-01-BR.
10. Harveson, R. M. 2011. A rare epidemic of sugar beet seedling rust in Nebraska. Proc. from the 36th Biennial Meetings of the Am. Soc. of Sugar Beet Technol., March 2-5, 2011, Albuquerque, NM. ASSBT, Denver, CO.
11. Newman, S. D., Gates, M., and Materne, M. 2006. Saltgrass. Plant Guide, USDA-NRCS, Washington, DC.
12. Oshima, N., Henderson, W. J., and Dickens, L. E. 1960. The occurrence of spinach rust in Colorado. Plant Dis. Reptr. 44:828-829.
13. Pool, V. W., and McKay, M. B. 1914. Puccinia subnitens on the sugar beet. Phytopathology 4:204-206.
14. Ramsey, G. B. 1943. Spinach rust in Colorado. Plant Dis. Reptr. 27:255.
15. Ramsey, G. B., and Smith, M. A. 1944. Orange rust, a market factor in Colorado spinach. Plant Dis. Reptr. 28:911-912.
16. Ramsey, G. B., Smith, M. A., and Wright, W. R. 1952. Orange rust, a serious market disease of Colorado spinach in 1952. Plant Dis. Reptr. 36:323-324.
17. Skoglund, L. G., and Brown, W. M. Red rust of spinach! Pest Alert, Vol. 16, No. 8. Coop. Ext., Colorado State Univ., Ft. Collins, CO.
18. Sherf, A. F., and Macnab, A. A. 1986. Vegetable Diseases and their Control, 2nd Edn. John Wiley and Sons, New York, NY.
19. Walker, J. C. 1952. Diseases of Vegetable Crops. McGraw-Hill, New York, NY.
