The sugar beet arose in Northern Europe as a product of breeding research. It was derived from the domesticated beet (Beta vulgaris L.), which previously had been developed in the second and third centuries A.D. by selection from wild beets native to the Mediterranean sea coasts and adjacent Northern Europe coastal areas. The relatively recently developed sugar beet crop plant has become well-known for acquiring new pathogens via native plants in any areas into which it is introduced. This phenomenon began in the mid 1800s and included the most important disease problems encountered early in European sugar beet production such as the cyst nematode and Rhizoctonia root rot.
The same relationship was noted after sugar beet cultivation was introduced into the Western Hemisphere. A new nematode disease was first discovered from Nebraska in the late 1940s that had never been documented prior to the arrival of the sugar beet in North America. In fact it has yet to be identified from anywhere else in the world where sugar beets are produced.
False Root-Knot Nematode
The false root-knot nematode (Nacobbus aberrans) has been labeled with several different names in the literature, including the Nebraska root-galling nematode, Cobb’s root-galling nematode, and the potato rosary nematode. Nematodes in the genus Nacobbus produce galls on the roots of their hosts that are similar in appearance to those caused by root-knot nematodes (Meloidogyne spp), providing the origin of this pathogen’s common name. Basing diagnosis on symptoms alone, the disease could easily be mistakenly attributed to Meloidogyne and therefore members of this species are familiarly known as false root-knot nematodes. The pathogen was named in honor of Nathan Augustus Cobb = Nacobbus, the pioneering USDA pathologist who is also considered the Father of American Nematology (Fig. 1).
 |
| Fig. 1. Nathan Augustus Cobb. (Photo: USDA) |
New Nematode Pathogen in North America
 |
|
| Fig. 2. Shadscale saltbush (Atriplex spp.), a common desert shrub native to the western United States. (Photo: Loren St. John, NRCS and Wikipedia Commons) |
|
Nathan Cobb was the first worker to observe specimens of what is now considered to be Nacobbus from Colorado in 1918, although he mistakenly documented them as Heterodera schactii (sugar beet cyst nematode). It is now known that the first species of Nacobbus was discovered and formally described by the prominent USDA nematologist Gerald Thorne. He found this nematode in 1935 infecting shadscale (Atriplex confertifolia), a native desert shrub (Fig. 2) on non-agricultural land in the foot hills of the Great Basin near Utah Lake, Utah, and named it Anguillulina aberrans.
The genus Nacobbus was eventually created in 1944, based on a new species found in southern California (N. dorsalis) and designated as the type species. Thorne’s Anguillulina aberrans was eventually moved into the genus Nacobbus, thereby becoming N. aberrans, and was distinguished from N. dorsalis by several key morphological features. Although N. dorsalis is the type species of Nacobbus, it is rare and considered of minor importance today. There have been no confirmed reports from any economic plant species, and has been found infecting only the redstem filaree (Erodium cicutarium) in California.
New Sugar Beet Disease in North America
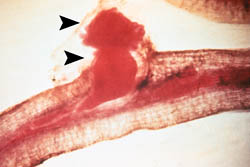
The false root-knot nematode was first isolated from sugar beets in 1949, near Mitchell, Nebraska by University of Nebraska plant pathologist Max Schuster, although the disease symptoms had been recognized and mistaken for infection by the root-knot nematode for many years prior to that. Infection can occur on young plants, often resulting in early-season loss of stand, as well as stunted and severely reduced foliar and root growth (Fig. 3) throughout the season. In mid to late season, severely infected plants wilt (Fig. 4) and exhibit pronounced yellowing during hot periods of the day (Fig. 5). The most conspicuous symptoms of N. aberrans on sugar beets are galls or swellings with a proliferation of lateral branches (Fig. 6) formed on mature taproots. These lateral roots may be studded with chains of irregular-shaped galls (Fig. 7), giving them a beaded appearance, with numerous small rootlets emerging from galls (Fig. 8).
 |
|
 |
| Fig. 3. Young sugar beet roots infected by N. aberrans. (Photo: R. M. Harveson) |
|
Fig. 4. Wilting symptoms of a sugar beet plant (right) infected by N. aberrans. (Photo: R. M. Harveson) |
 |
|
 |
| Fig. 5. Yellowing of sugar beet plants in a field infested with N. aberrans near harvest. Note also the large gaps in the foreground due to previous plant mortality. (Photo: R. M. Harveson) |
|
Fig. 6. Mature sugar beet roots at harvest exhibiting the proliferation of lateral roots off the taproot, characteristic of the false root-knot nematode (N. aberrans). (Photo: R. M. Harveson) |
 |
|
 |
| Fig. 7. Mature sugar beet root exhibiting galls on lateral root branches, characteristic of the false root-knot nematode (N. aberrans). (Photo: R. M. Harveson) |
|
Fig. 8. Close-up image showing small rootlets emerging from galls of N. aberrans-infected sugar beet taproot. (Photo: R. M. Harveson) |
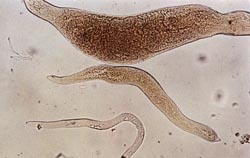
This disease was first described, characterized, and published from western Nebraska in 1956 by Schuster and Thorne. After their comprehensive investigations involving the pathogen’s distribution, host range, life cycle, and pathology were completed, it was concluded that the sugar beet pathogen from Nebraska was distinct from the two species previously described as N. dorsalis and N. aberrans, and newly named N. batatiformis, based on the shape of the mature female. The female’s body at maturity was tapered at both ends, resembling a sweet potato (Ipomoea batatas) (Fig. 9). This morphological feature further distinguished it from other gall-producing nematodes like Meloidogyne spp. that are characterized by pear-shaped bodies with a rounded posterior end (Fig. 10). However, N. batatiformis was later merged with N. aberrans, retaining the latter name.
 |
|
 |
| Fig. 9. Sweet potato-shaped mature female of false root-knot nematode (top) with several stages of juvenile forms (below). (Photo: M. L. Schuster, University of Nebraska) |
|
Fig. 10. Pear-shaped body of root-knot (Meloidogyne spp.) nematode female within infected root (lower arrow). Note the eggs exuding out of the nematode’s body in a gelatinous mass (top arrow). (Photo: N. A. Mitkowski and G. S. Abawi) |
Distribution
| |
|
| |
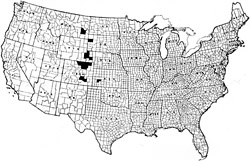
Fig. 11. Distribution of false-root knot incidence within the Great Plains of the United States as a result of an early nematode survey. (Photo: F. E. Caveness) |
A regional survey of sugar beet production in the late 1950s determined that although disease incidence was not as widespread as that of the sugar beet cyst nematode, the false root-knot pathogen was still identified from 16 counties in 6 states throughout the Great Plains, including Colorado, Kansas, Montana, Nebraska, South Dakota, and Wyoming (Fig. 11). Infestation of sugar beet fields by this nematode today is still limited to the central high plains east of the Rocky Mountains. Another earlier survey conducted by Schuster in western Nebraska exclusively over two years found the nematode infesting one third (32%) of 125 sugar beet fields monitored.
It is still considered a potentially serious pest in this region due to its widespread presence and because of the potential for serious yield reductions when infection does occur. Presently, Nebraska and Wyoming are the states most commonly and severely affected by this pest, with losses in sugar beets crops ranging from 10 to 20% being documented.
Pathogen Host Range
In the United States, N. aberrans is primarily an economic problem on sugar beet, but the pathogen can also affect numerous other vegetable hosts such as carrot, pea, lettuce, tomato, and numerous species in the mustard (broccoli, cabbage, rutabaga, radish, and turnip) and cucurbit (pumpkin, cucumber) families. Fortunately, with the exception sugar beet, field crops grown in the central high plains of the United States under irrigation, including potatoes, corn, dry beans, sunflower, alfalfa, wheat, and barley, are not highly susceptible. Because of the broad host range on horticultural crops, the disease can still be problematic for city residents with backyard gardens, particularly with root vegetables like beets, turnips, radishes, and carrots.
Interestingly, the most commonly identified noneconomic hosts for the nematode in western Nebraska were determined to be three species of cacti, including two species of the prickly pear, Opuntia (O. tortispina and O. fragilis), and the spiny star cactus, Escobaria vivapara. All of these species are native to the central high plains region and other locations throughout western North America. Nematode colonies isolated from both cacti and sugar beets each resulted in reciprocal infections with an identical colony reproduction and morphology. The false root-knot nematode has also been found commonly throughout the region infecting indigenous weed species, including various mustards (Brassica spp.), common lambsquarters (Chenopodium album) (Fig. 12), and Kochia (Bassia scoparia) (Fig. 13), as well as the introduced species common purslane (Portulaca oleracea), puncturevine (Tribulus terrestris), and Russian thistle (Salsola tragis).
 |
|
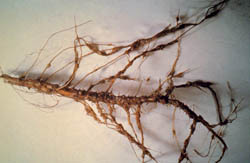 |
| Fig. 12. Gall formation on N. aberrans-infected lambsquarters (Chenopodium album) root. (Photo: R. M. Harveson) |
|
Fig. 13. Gall formation on N. aberrans-infected Kochia (Bassia scoparia) root. (Photo: E. D. Kerr, University of Nebraska) |
R. N. Inserra and colleagues determined from greenhouse studies that the Chenopodiacious weed, Kochia appeared to be more tolerant to the pathogen than sugar beet, suffering less damage in response to infection. Although these studies indicate that Kochia may be a poorer host for the nematode, they also provide evidence that this host can serve as a survival mechanism for N. aberrans, extending its longevity and maintenance in soils.
Taxonomic Confusion
The taxonomic history of N. aberrans has been controversial. Although most nematologists today recognize two primary species, N. aberrans and N. dorsalis, it is now becoming increasingly evident that a revision of the taxon is urgently needed as its species differences have not been fully resolved. This is further illustrated by reports in the literature over the years of three additional species from wide-ranging, contrasting locations, including the previously mentioned N. batatiformis from Nebraska, N. serendipiticus from greenhouse tomato production in Great Britain, and N. serendipiticus bolivianus from potatoes in Bolivia. In 1969, S. A. Sher transferred the latter three species into the heterogeneous N. aberrans, further complicating the taxonomy of this nematode.
However, at present, it is still generally accepted that the false root-knot nematode can be separated into at least three distinct groups or pathotypes, based on different host ranges and DNA characteristics. These pathotypes include a strain from the highland Andean regions of Argentina, Bolivia, Chile, and Peru that infects potatoes but not peppers. A bean strain from Mexico has also been found that damages beans, tomatoes, and peppers, but not sugar beet or potato. Lastly, the sugar beet strain previously discussed has been well characterized biologically and documented from the Great Plains of the United States. It infects sugar beets and many common vegetable crops, but not potato. Recent analyses of ITS sequences appear to confirm the validity of separating the pathogen into these specific subgroups, but no studies have compared populations from the Great Plains, Mexico, and Andes groups simultaneously.
Conclusions
The ability of the North American sugar beet pathotype of N. aberrans to parasitize nonindigenous host species makes it quite unique when compared to the two pathotypes from Mexico and South America that infect primarily the New World solanaceous (potato, peppers, and tomato) and leguminaceous (bean) crops.
It is clear that the genus Nacobbus (false root-knot nematode) is distinct from the various Meloidogyne species (root-knot nematodes) with which it has often been confused, and the species is also unquestionably endemic to the Americas. It has furthermore been demonstrated to occur in multiple, diverse areas of the Western Hemisphere, but is absent from any other locale in the world. The presence of the sugar beet strain of N. aberrans (initially named N. batatiformis) occurring for more than a half century on native cacti (Fig. 14) from uncultivated rangeland in Scotts Bluff, Sioux, and Morrill counties of the western Nebraska Panhandle has provided convincing evidence that this nematode was indigenous to this region, but then adopted the sugar beet as its preferred host after the crop was introduced into North America.
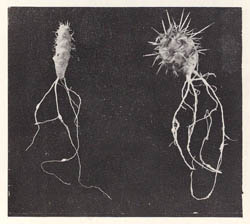 |
| Fig. 14. Gall formation on N. aberrans-infected prickly pear cactus (Opuntia spp.). (Photo: M. L. Schuster, R. Sandstedt, and L. W. Estes, University of Nebraska) |
References and Further Reading
1. Altman, J., and Thomason, I. 1971. Nematodes and their control. Pages 335-370 in: Advances in Sugarbeet Production: Principles and Practices. R. T. Johnson, J. T. Alexander, G. E. Rush, and G. R. Hawkes, eds. Iowa State University Press, Ames, IA.
2. Bennett, C. W. 1973. A consideration of some of the factors important in the growth of the science of plant pathology. Ann. Rev. Phytopathol. 11:1-11.
3. Caveness, F. E. 1959. Distribution of cyst- and gall-forming nematodes of sugar beets in the United States. J. Am. Soc. Sugar Beet Technol. 10:544-552.
4. Clark, S. A. 1967. The development and life history of the false root-knot nematode, Nacobbus serendipiticus. Nematologica 13: 91-101.
5. EPPO/CABI. 1996. Nacobbus aberrans. In: Quarantine Pests for Europe, 2nd Ed. I. M. Smith, D. G. McNamara, P. R. Scott, and H. Holderness, eds. CAB International, Wallingford, UK.
6. Harveson, R. M. 2008. False root-knot nematode. Coop. Ext. Serv. NebGuide G1857, University of Nebraska, Lincoln.
7. Harveson, R. M., Panella, L., and Lewellen, R. T. 2009. History of beet production and usage. Pages 1-2 in: Compendium of Beet Diseases and Pests. R. M. Harveson, L. E. Hanson, and G. L. Hein, eds. American Phytopathological Society, St. Paul, MN.
8. Ibrahim, S. K., Baldwin, J. G., Roberts, P. A., and Hyman, B. C. 1997. Genetic variation in Nacobbus aberrans: An approach toward taxonomic resolution. J. Nematol. 29:241-249.
9. Inserra, R. N., Griffin, G. D., Vovlas, N., Anderson, J. L., and Kerr, E. D. 1984. Relationship between Heterodera schachtii, Meloidogyne hapla, and Nacobbus aberrans on sugarbeet. J. Nematol. 16:135-140.
10. Inserra, R. N., Di-Vito, M., and Ferris, H. 1984. Influence of Nacobbus aberrans densities on growth of sugarbeet and Kochia in pots. J. Nematol. 16:393-395.
11. Inserra, R. N., Chitambar, J. J., Chitwood, D. J., and Handoo, Z. 2005. The potato pathotype of the false root-knot nematode, Nacobbus aberrans. A List of Exotic Nematode Plant Pests of Agricultural and Environmental Significance to the United States. Online. University of Nebraska-Lincoln, Society of Nematologists, and USDA-APHIS. http://nematode.unl.edu/projectpest.htm.
12. Powers, T. 2004. Nematode molecular diagnostics: From bands to barcodes. Ann. Rev. Phytopathol. 42:367-383.
13. Manzanilla-Lopez, R. H., Costilla, M. A., Doucet, M., Franco, J., Inserra, R. N., Lehman, P. S., Cid del Prado-Vera, I., Souza, R. M., and Evans, K. 2002. The genus Nacobbus Thorne & Allen, 1944 (Nematoda: Pratylenchidae): Systematics, distribution, biology and management. Nematropica 32:149-227.
14. Reid, A., Mazanilla-Lopez, R. H., and Hunt, D. J. 2003. Nacobbus aberrans (Thorne, 1935) Thorne and Allen, 1944 (Nematoda: Pratylenchidae); a nascent species complex revealed by RFLP analysis and sequencing of the ITS-rDNA region. Nematology 5:441-451.
15. Sauer, J. D. 1994. Historical geography of crop plants. CRC Press, Boca Raton, FL.
16. Schuster, M. L., and Thorne, G. 1956. Distribution, relation to weeds, and histology of sugar beet root galls caused by Nacobbus batatiformis, Thorne and Schuster. J. Am. Soc. Sugarbeet Technol. 9:193-197.
17. Schuster, M. L., Sandstedt, R., and Estes, L. W. 1964. Starch formation induced by a plant parasitic nematode. Science 143:1342-1343.
18. Schuster, M. L., Sandstedt, R., and Estes, L. W. 1965. Host-parasite relations of Nacobbus batatiformis and the sugar beet and other hosts. J. Am. Soc. Sugarbeet Technol. 13:523-537.
19. Sher, S. A. 1970. Revision of the genus Nacobbus Thorne and Alle, 1944 (Nematoda: Tylenchoidea). J. Nematol. 2:228-235.
20. Thorne, G. 1935. The sugar beet nematode and other indigenous nemic parasites on shadscale. J. Agric. Res. 51:509-514.
21. Thorne, G., and Schuster, M. L. 1956. Nacobbus batatiformis, n. sp. (Nematoda: Tylenchidae), producing galls on the roots of sugar beet and other plants. Proc. Helminthol. Soc. Wash. 23:128-134.
22. Thorne, G. 1961. Principles of Nematology. Mcgraw Hill, New York, NY.
23. Weihing, J. L., and Kerr, E. D. 1968. Nebraska root-gall nematode of sugar beets. Ext. Circ. 68-1847, Coop. Ext. Serv., University of Nebraska-Lincoln.
