Introduction
Curly top of sugar beet was among the first plant viral diseases recognized (with tobacco mosaic) in the late nineteenth century. It was also the second disease shown to be associated with a leafhopper vector, the beet leafhopper (Circulifer tenellus) (Fig. 1), the dwarf disease of rice from Japan being the first. Since its discovery, beet curly top has likely been the most intensively studied of all the leafhopper-transmitted virus diseases owing to its past and continuing impact on multiple economically important crops.
 |
| Fig. 1. Beet leafhopper. (Courtesy L. D. Godfrey; Reproduced, by permission, from Compendium of Beet Diseases and Pests) |
Discovery of Curly Top
Knowledge of curly top’s early history in the United States is limited owing to its sharing similar symptoms with other diseases and disorders. According to C. O. Townsend, George G. Hedgecock with the USDA reported that, on several occasions before 1888, he observed damaging outbreaks of a disease presumed to be curly top from garden beets in Nebraska. In 1897, George Austin stated that the Utah Sugar Company suffered overwhelming losses near Lehi, UT from an unidentified disorder resembling curly top.
The disease also was noticed in California in the 1890s, with severe losses reported from the newly emerging sugar beet industry in that state. By 1900, it had been noted throughout all western states where sugar beets were grown, suggesting it was already widely distributed before it was first recognized or studied as a distinct disease.
This hypothesis is conceivable because the first description of the beet leafhopper on sugar beets was made from the western slope of Colorado in 1895 by C. P. Gillette and C. F. Baker. As early as 1905, E. D. Ball suspected the beet leafhopper in the spread and development of sugar beet curly top disease in Utah. However, he did not attribute the appearance of the disease to any viral or pathogenic entity, figuring that the damage was directly caused by the feeding injury induced by the insect.
|
 |
|
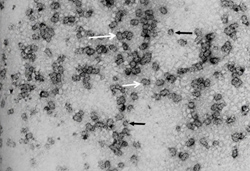
Fig. 2. Electron micrograph of curly top virus “geminate” particles. Most are twinned, in groups or clusters of two particles (white arrows), while single particles are depicted with black arrows. (Courtesy F. Hayes; Reproduced from Phytopathol. 64:136-139) |
It was not until 1915 that the relationship between the vector and the virus in causing disease was irrefutably established by P. A. Boucquet and W. J. Hartung. They showed that after leafhoppers taken from the wild with no previous contact with sugar beets were allowed to feed on beets, no disease ensued. But when they were allowed to feed on beets with symptoms of curly top for 3–7 days, curly top symptoms appeared on previously healthy beets after leafhoppers were transferred and granted time to feed.
The discovery of curly top transmission by leafhoppers strongly suggested a viral etiology; however, the pathogen’s identity was not formally established until the 1970s. The typically low concentration of virus particles within infected plants made isolation and purification difficult. Therefore, it was not until 1974 that a virus was formally proven to be the cause of the disease, after advancements in virus purification were made and the characteristic twinned particles of geminiviruses were observed with electron microscopy (Fig. 2).
Characteristics of Leafhopper-Transmitted Virus Diseases
Virus diseases transmitted by leafhoppers were the first group of plant diseases noted to be associated with an insect vector, and most diseases in this group possess a number of common characteristics. They are generally transmitted in a persistent manner (in which the virus continues to exist within the insect’s body). Leafhopper-transmitted diseases also exhibit a high degree of specificity for their host plants and a distinctly close biological relationship between the vector and pathogen.
All viruses in this group cause a general disruption of the plant vascular system, usually the phloem. They are not readily transmitted mechanically nor are they seedborne, most likely due to the nature of the virus’s presence within phloem tissues.
The Pathogens
Curly top is actually caused by a group of several closely related viruses that vary substantially in host range and virulence, and is the only currently known plant virus disease caused by pathogens consisting of genomes of single-stranded DNA. Taxonomically they are in the genus Curtovirus (family Geminiviridae), and the type member of the subgroup II of the group collectively known as the geminiviruses. All geminiviruses contain the distinctive paired or twin particles, also referred to as geminate, each of which is the result of partial fusion of two isometric particles (Fig. 2).
Three Different Strains
Early workers recognized and distinguished several distinct strains based on differences in symptom severity long before the nature of the virus and its tremendous diversity was established. In the mid-1980s an infectious clone of the curly top pathogen was developed from an isolate originally found near Modesto, CA in the 1930s. This original isolate was kept alive by serial transfer within the greenhouse by C. W. Bennett and J. E. Duffus (USDA-ARS, Salinas, CA) for more than 50 years. Three other isolates similarly maintained in greenhouses were also molecularly characterized by cloning and sequencing. Based on DNA sequence characters and disease severity differences on sugar beets, these strains have now been separated into three distinct species of geminiviruses, including CFH, Worland, Cal/Logan, and various genotypic variants of these three strains. CFH is now called Beet severe curly top virus (BSCTV), Worland is known as Beet mild curly top virus (BMCTV), and Cal/Logan is now referred to as Beet curly top virus (BCTV).
The presence of BCTV was traditionally associated with California agricultural production, but now appears to be rare in any of the sugar beet-producing regions. Curly top surveys done in 1994 and 1995 throughout the United States determined that BSCTV and BMCTV were more widely distributed and commonly identified, with very few isolates of BCTV being detected at all. Another survey of the western United States a decade later found very similar results. Based on these studies, most damage today is thought to be due to BSCTV and BMCTV.
Host Range and Transmission
Curly top has an extraordinarily large number of wild and economic hosts (more than 300 species from at least 44 families). Economic hosts include sugar and table beet, Swiss chard, spinach, tomato, pepper, bean, flax, and numerous cucurbits including melons, squash, pumpkin, and cucumber. The beet leafhopper also has an extensive host range and transmits the viral pathogen very efficiently in a persistent manner. The insect is capable of producing three or more generations per season, and moving long distances in wind currents from its breeding grounds to cultivated fields.
The beet leafhopper cannot transmit the viral pathogen immediately after feeding but requires at least a four-hour latent period after acquisition; afterward, inoculation can occur in as little as one minute of feeding. The most efficient transmission is accomplished after a 48-hour acquisition period. The insect can carry and transmit all curly top strains simultaneously, while also remaining viruliferous for long periods of time (more than 3 months in some instances), consistent with other leaf hopper-transmitted virus diseases.
Symptoms
Beets infected young often die quickly. Plants that are infected later and survive are characterized by yellowing and death of mature leaves. Newer leaves are smaller and more numerous compared to healthy plants while also tending to roll inward and upward (Fig. 3) with blister-like swellings forming on veins on the underside of leaves, resembling small galls (Fig. 4).
Due to the pathogen’s confinement within phloem tissues, necrosis, degeneration, and death of the periderm and phloem cells adjacent to sieve tubes ensue. Phloem necrosis also is easily observed as dark concentric rings or linear streaks in cross section or longitudinal section of the taproot, respectively (Fig. 5).
 |
|
 |
| Fig. 3. Curly top symptoms on sugar beet exhibiting upward and inward leaf curling. (Photo by R. M. Harveson) |
|
Fig. 4. Curly top symptoms on sugar beet: enations on underside of leaves. (Photo by R. M. Harveson) |
 |
| Fig. 5. Phloem discoloration in sugar beet roots from curly top infection. (Photo by Eubanks Carsner, USDA) |
Vector Behavior and Disease Epidemiology
Before the development of commercialized agriculture in the western United States, the native flora did not support large, economically damaging populations of the beet leafhopper. Conditions favoring the insect modified this dynamic with the changes in agriculture, including sporadic, intermittent farming and overgrazing of rangeland. This series of factors resulted in the disappearance of previously stable plant populations that were replaced by various winter and summer annual weed species. The weed hosts that succeeded the original plant species included mustards (Brassica spp.), Russian thistle (Salsola tragus), filaree (Erodium spp.), and Plantago spp. These widespread weeds became the preferred leafhopper hosts resulting in more frequent and severe epidemics.
In the fall as crops senesce, the leafhopper vector moves to breeding areas in the foothills away from cultivated fields, overwintering on various biennials and perennials. Winter rainfall results in the germination and emergence of several annuals that serve as sites of leafhopper reproduction. The leafhopper moves to these annual plant species and lays eggs. In spring as plants in breeding areas dry, the leafhopper moves back to agricultural lands. The severity of infection depends on climatic factors affecting the presence of weed hosts, the prevalence and severity of the pathogen, and the reproductive capacity and migration of leafhoppers.
Economic Effects of Curly Top
Although crop failures are rare today, curly top is still a potentially problematic issue for sugar beet production in several areas of the western United States The disease has been for the most part confined to the western half of the North American continent and is generally more severe west of the continental divide. It does occur sporadically in the beet-growing areas of the high plains east of the Rocky Mountains, but is not often an economically limiting factor.
After 1900 curly top rapidly emerged as a threat to American sugar beet production in the west. It crippled and seriously threatened the survival of the new sugar beet industry in the first three decades of the 20th century. This one disease was responsible for the closure of numerous processing plants across this growing region, in a similar manner to the impact of the cyst nematode on the European continent in the latter half of the 19th century. Its damage also directly contributed to wild fluctuations in sugar prices due to varying supply and demand relationships, causing the cultivation of beets to constantly move to new locations, leaving others abandoned. As an example, the worst year for California’s Spreckles Sugar Company occurred in 1926. Following this year, the entire company very nearly closed and declared bankruptcy as a direct result of curly top, after only 7500 acres were harvested and processed.
Breeding for Resistance
Because of successive crop failures from curly top throughout the intermountain west of the United States, the USDA began breeding for resistance in 1925 with several approaches: searching for new genes from wild beets relatives, evaluating and selecting individual types within existing commercial varieties, and establishing field plots in locations expected to be exposed to severe infection (Fig. 6) for optimal resistance evaluations. By 1929, Congress had appropriated additional funds for continuing this line of research.
 |
| Fig. 6. Resistance evaluations under severe curly top infection. Rows 84, 85, and 86 are hybrids of sugar beet and the wild species, B. maritima. Compare results to two flanking rows on each side consisting of commercial varieties. (USDA photo) |
Eubanks Carsner, USDA plant pathologist, was assigned the task of investigating this disease, and he began by selecting individual plants exhibiting resistance from fields with high levels of disease pressure, a system referred to as mass selection. The accumulated seed (approximately 100 pounds) produced between 1925 and 1929 from surviving plants from this method was first tested in 1930 in Idaho under conditions of severe pressure (Fig. 7). The surviving progeny from these field tests eventually became the variety US 1, which averaged 5.5 tons higher than susceptible European varieties. Steps were then taken to increase seed of this line as a source of resistance to curly top from the unused seed stocks.
Although the use of this new variety was immediately recognized as only temporary, it was still released as the first curly top-resistant cultivar for commercial production in 1933 due to the dire situation the disease posed to the industry. US 1 was quickly replaced by other superior lines (US 33 and 34) that were created from reselections from US 1 (Fig. 8), both of which were more resistant than their parent varieties. As an example of this dramatic improvement, 85% of planted sugar beet acreage in 1934 was abandoned with the average national yield registering a paltry 4.9 tons/acre. By 1941, 97% of the acreage was harvested and processed with national yields of 13.5 tons/acre being achieved. By the mid-1940s, the disease no longer posed a serious problem to the beet sugar industry. For this achievement, Carsner and his staff received the Superior Service Award of the U.S. Department of Agriculture in 1947.
 |
|
 |
| Fig. 7. The initial test using U.S. 1 under severe disease conditions in Idaho in 1930. Note the plot to the right of the man standing compared to adjacent plots. It consists of 4 rows of U.S. 1. (Photo by Eubanks Carsner, USDA) |
|
Fig. 8. Curly top resistance in sugar beet. The plants in the row on the right were the progeny obtained from a plant selected for resistance. Compare to the adjacent rows to the left consisting of commercial varieties. (Photo by Eubanks Carsner, USDA) |
Sugar Beet Seed Production
In the early 1900s, the sugar beet was unique among most American crops in its almost complete dependence upon Europe as a source for seed each season. At this time, the vast majority of all seed used in the United States was produced in Europe. There were two major factors that convinced Americans that they needed to establish a domestic seed industry – the First World War and curly top.
Beginning about 1915, owing to escalating war conditions in Europe, the quality of seed either deteriorated or seed became generally unavailable, which began illustrating the need for an American sugar beet seed industry. Many individuals within the industry recognized and understood the potential benefits for producing seed domestically. Nevertheless it still took some time before everyone finally came to this realization, even though work done 20+ years earlier had determined that success in this venture was definitely feasible.
Early Experiments in Seed Production in the United States
|

|
|
|
Fig. 9. Harvey Washington Wiley. USDA Yearbook of Agriculture, 1962. |
|
Successful sugar beet seed production on an experimental basis was reported by Harvey W. Wiley (Fig. 9), chief of the U.S. Department of Agriculture’s Bureau of Chemistry, as early as 1887. The first serious effort to grow seed in the United States occurred in Schuyler, Nebraska, in 1891. Wiley continued these studies for a number of years, achieving steady and continuous progress in sugar beet seed improvement. The USDA conducted additional beet seed production experiments in the Pacific northwest at Fairfield, Washington, with similar success, and the process was further tested in other states ranging from Michigan to Idaho under highly diverse soil and environmental conditions. The combined results of these studies provided abundant proof that domestic seed production could be achieved satisfactorily, and also suggested even better results could be secured via creating and increasing new seed under regionally adapted conditions for use in different growing areas.
Wiley’s efforts not only proved that successful production was possible but that American-grown seed was actually of higher quality, producing higher sucrose yields than the seed imported from Europe. Domestically-produced seed was also superior in germination and capable of producing larger and better roots. Based on these studies, the USDA recommended that this proactive approach should continue and began producing seed regionally adapted to the various American sugar beet-producing regions.
Domestic Seed Production Revisited
Wiley’s preliminary investigations took on a greater significance and became increasingly important when the European seed availability was cut off due to WWI. Sugar companies were then forced to revisit the concept of seed production and because their impromptu efforts were hastily implemented, success was not immediately realized. Yields ranged from only a few hundred to 2000 pounds per acre with 800 pounds as an average.
After WWI, the industry returned to the practice of importing seed from Europe. We should have learned previously from the disastrous effects to production suffered as a result of the prior dependence upon European sources of seed. Unfortunately, this lesson was soon forgotten because of greater convenience and lower costs. Approximately 15,000,000 pounds of seed were imported annually from Europe between 1920 and 1933. However, it was curly top that finally provided the most dramatic and convincing reason for establishing an independent sugar beet seed industry in the United States. Curly top did not exist in Europe, thus the varieties grown in the United States from European seed sources were all highly susceptible to the disease.
Botany of the Sugar Beet
The sugar beet is a biennial plant and vegetative growth during the first year is required for bulking sucrose in the roots for the following year’s reproductive growth. Sugar beets tend to behave as a perennial if flowering is not induced, and therefore the induction of flowering is required for seed production (Fig. 10). Flowering is stimulated after a period of cool temperatures and long nights, known as vernalization. By the European method of seed production, roots were harvested and removed from the field by hand in the fall, and placed in a 4°C cooler for 12–16 weeks to induce vernalization. The following spring, they were replanted back in the field. Flowering then began within five weeks after removing the plants from vernalization, followed by seed production and maturation later that summer.
 |
| Fig. 10. Bolting (flowering) sugar beet plant producing seed on long stalks. (Photo by R. M. Harveson) |
New Method of Seed Production
An alternative method better suited for producing seed in the United States was first tested in 1922 with the USDA collaborating with the New Mexico Agricultural Experiment Station (Fig. 11). Producing seed in the American Southwest took advantage of the mild winters, enabling roots to overwinter in the field. Plantings made in early fall were left in the field over the winter before being harvested mechanically as seed the following summer (Fig. 12). This reduced the overall production expenses compared with the European method which entailed high costs of labor required to remove roots from fields in fall, store them over the winter, and replant them the following spring.
 |
|
 |
| Fig. 11. A field planted for seed production for the overwintering method near Las Cruces, NM. (Photo by G. H. Coons, USDA) |
|
Fig. 12. Harvesting sugar beet seed. (Photo by C. O. Townsend, USDA) |
Furthermore, yields were consistently much higher than those from conventional European practices. Similar experiments were successfully conducted in frost-free areas of southern Utah and California. By this method, the curly top-resistant variety US 1 was successfully produced and increased, making it rapidly available to growers. These alternative procedures generated enough seed by 1934 that 55,000 acres of the resistant variety were planted that year in those curly top-problematic areas of the west.
The widely successful creation of curly top-resistant varieties and their increase rejuvenated the sugar beet industry in many production areas that were abandoned earlier owing to repeated losses using the curly top-susceptible European varieties.
Curly Top’s Enduring Legacy
The curly top disease has served as an important stimulus for spurring new advancements in sugar beet production in the western half of North America, including the production of locally adapted, disease-resistant varieties as this was not being addressed by European companies. The development of locally adapted cultivars, in turn, then created a need for a domestic seed industry. Eubanks Carsner was one of the pioneers in developing our present sugar beet seed industry. He had the foresight to envision high-quality sugar-beet seed being grown in the Pacific Northwest by the overwintering method. Following his recommendations, a domestic company was established at Salem, Oregon, and most of our seed today is produced in the Willamette Valley of that state.
Conclusions
The successful production of curly top-resistant beet seed showed the need for producing varieties adapted to local, diverse, beet-growing regions. This complete reversal in policy for utilizing European seed exclusively resulted in the development of new a breeding program and domestic seed industry that has been credited with saving the fledgling sugar beet industry in the western United States, and is still functioning effectively today.
References and Further Reading
Annand, P. N. 1931. Beet leafhopper’s annual migrations studied in desert breeding areas. Pages 114-116 in: Yearbook of Agriculture. USDA, Washington, DC.
Ball, E. D. 1906. The beet leaf-hopper (Eutettix tenulla). Utah Agr. Exp. Sta. Ann. Rpt. 16 (1904-1905).
Bennett, C. W. 1967. Epidemiology of leafhopper-transmitted viruses. Ann. Rev. Phytopathol. 5:87-108.
Bennett, C. W. 1971. The Curly Top Disease of Sugarbeet and Other Plants. Monograph 7. American Phytopathological Society, St. Paul, MN.
Bennett, C. W. 1973. A consideration of some of the factors important in the growth of the science of plant pathology. Ann. Rev. Phytopathol. 11:1-11.
Boncquet, P. A., and Hartung, W. J. 1915. The comparative effect upon sugar beets of Eutettix tenulla Baker from wild plants and from curly top beets. Phytopathol. 5:348-349.
Brandes, E. W., Townsend, C. O., Yoder, P. A., Sherwood, S. F., Washburn, R. S., Arner, G. B. L., Baker, O. E., Stevens, F. C., Chittenden, F. H., and Langworthy, C. F. 1924. Sugar. Pages 151-228 in: Yearbook of Agriculture. USDA, Washington, DC.
Carsner, E. 1924. Studies on curly top disease of the sugar beet. J. Agric. Res. 28:297-320.
Carsner, E. 1927. Sugar beet disease called curly top limits production. Pages 603-605 in: Yearbook of Agriculture. USDA, Washington, DC.
Carsner, E., and Piemeisel, R. L. 1931. Sugar beet curly top’s spread aided by vast increase in host weeds. Pages 491-493 in: Yearbook of Agriculture. USDA, Washington, DC.
Coons, G. H. 1928. Sugar-beet disease control progressing toward solution. Pages 557-559 in: Yearbook of Agriculture. USDA, Washington, DC.
Coons, G. H., Stewart, D., and Elcock, H. A. 1931. Sugar-beet strains resistant to leaf spot and curly top. Pages 493-496 in: Yearbook of Agriculture. USDA, Washington, DC.
Coons, G. H. 1936. Improvement of the sugar beet. Pages 625-656 in: Yearbook of Agriculture. USDA, Washington, DC.
Coons, G. H., Owen, F. V., and Stewart, D. 1955. Improvement of the sugar beet in the United States. Adv. Agron. 8:89-135.
EPPO/CABI. 1982. Beet curly top hybrigeminivirus. Quarantine Pests for Europe, 2nd ed. I. M. Smith, D. G. McNamara, P. R. Scott, and H. Holderness, eds. CAB International, Wallingford, UK.
Gillette, C. P., and Baker, C. F. 1895. A preliminary list of Hemiptera of Colorado. Colorado Agr. Exp. Sta. Bull. 31.
Harveson, R. M., Panella, L., and Lewellen, R. T. 2009. History of beet production and usage. Pages 1-2 in: Compendium of Beet Diseases and Pests. R. M. Harveson, L. E. Hanson, and G. L. Hein, eds. American Phytopathological Society, St. Paul, MN.
Lewellen, R. T., Panella, L., and Harveson, R. M. 2009. Botany of the beet plant. Pages 2-3 in: Compendium of Beet Diseases and Pests. R. M. Harveson, L. E. Hanson, and G. L. Hein, eds. American Phytopathological Society, St. Paul, MN.
Oman, P. W. 1948. Notes on the beet leafhopper, Circulifer tenellus (Baker) and its relatives (Homoptera: Cicadellidae). J. Kansas Entomol. Soc. 21:10-14.
Panella, L., Lewellen, R. T., and Harveson, R. M. 2009. Breeding for disease and insect resistance. Pages 3-5 in: Compendium of Beet Diseases and Pests. R. M. Harveson, L. E. Hanson, and G. L. Hein, eds. American Phytopathological Society, St. Paul, MN.
Stefferud, A. 1962. Men and Milestones. Yearbook of Agriculture. USDA, Washington, DC.
Stenger, D. C., and McMahon, C. L. 1997. Genotypic diversity of beet curly top virus populations in the western United States. Phytopathology 87:737-744.
Strausbaugh, C. A., Wintermantel, W. M., Gillen, A. M., and Eujayl, I. A. 2008. Curl top survey in the western United States. Phytopathology 98:1212-1217.
Townsend, C. O. 1908. Curly-top, a disease of the sugar beet. U.S. Dept. Agr. Bur. Plant Ind. Bull. 122.
Townsend, C. O. 1917. The present status of the sugar-beet seed industry in the United States. Pages 399-410 in: Yearbook of Agriculture. USDA, Washington, DC.
Tracy, J. E. W. 1904. Sugar-beet seed breeding. Yearbook of Agriculture. USDA, Washington, DC.
Wintermantel, W. M. 2009. Curly top. Pages 51-53 in: Compendium of Beet Diseases and Pests. R. M. Harveson, L. E. Hanson, and G. L. Hein, eds. American Phytopathological Society, St. Paul, MN.
Wintermantel, W. M., and Kaffka, S. R. 2006. Sugar beet performance with curly top is related to virus accumulation and age at infection. Plant Dis. 90:657-662.
Wisler, G. C., and Duffus, J. E. 2000. A century of plant virus management in the Salinas Valley of California, ‘East of Eden’. Virus Res. 71:161-169.
