Introduction
Bacterial wilt of dry beans (Phaseolus vulgaris L.), caused by Curtobacterium flaccumfaciens pv. flaccumfaciens (Cff) (formerly Corynebacterium flaccumfaciens), has been a sporadic but often serious production issue since first being reported from South Dakota in 1922. By the 1940s it became one of the most problematic bacterial diseases in the United States, particularly throughout the irrigated high plains and Midwest. Wilt then persisted through the early 1970s as an endemic and economically important production constraint, presumptively through seedborne transmission and dissemination, before essentially disappearing by the early 1980s. The disease then only periodically appeared in seed, but had little detectable effect on yields after the implementation of crop rotation and seed sanitation practices.
Over the last decade, the author has comprehensively studied the history of this disease and personally investigated the new outbreaks throughout the Central High Plains of western Nebraska, northeastern Colorado, and southeastern Wyoming. Concurrently, the author has further documented some of the pathogen’s distinctive features that distinguish it from other more familiar bacterial pathogens affecting dry beans. Based on these observations, the author has postulated that the disease’s re-emergence in this region can be explained by a combination of factors, some of which we have had no control over while others are inarguably of our own doing.
Re-emergence of Wilt in Dry Bean Production
In 1995 and 2002, new reports concerning bean wilt were published from North Dakota and Alberta, Canada respectively, after an apparently extended hiatus in North America. Both accounts described finding orange and yellow discolored seeds from cull beans that were obtained from growers and processing plants after harvest, but no mention was ever made of seeing symptomatic plants in the field or experiencing yield reductions.
Late in 2003, the disease was again identified from two fields planted to great northern beans in Scotts Bluff Co., Nebraska, representing the first notice in this region for more than 20 years. Since that time the disease has been frequently and regularly observed, and determined to be widely distributed over the Central High Plains, particularly from western Nebraska.
Furthermore, and in contrast to the new reports from North Dakota and Alberta, severe disease symptoms in fields prior to harvest have also been routinely documented (Fig. 1) accompanied by definitive, measurable economic losses. For example, several heavily infested fields in Nebraska have suffered yield reductions approaching 65-70% less than would be expected of a typical crop. Other fields have been destroyed without being harvested. Numbers of discolored seeds collected from surviving but infected plants were found to average 25-30% of the total produced for each plant (Fig. 2), while the incidence of discolored great northern seeds harvested from entire fields approached 10% in some instances.
 |
|
 |
|
Fig. 1. Great Northern dry bean field in Nebraska severely affected by wilt. Note the large gaps without plants, indicating early infection of seedlings that did not survive. |
|
Fig. 2. Yield of one infected dry bean plant showing staining of surviving, mature seeds.
Note that not all seeds become infected. |
Field symptoms in Nebraska consisted of leaf wilting and death during periods of warm, dry weather or periods of moisture stress (Fig. 3), and very distinctive interveinal, necrotic lesions surrounded by wavy, bright yellow borders, symptoms referred to as "firing" (Fig. 4). Affected fields throughout the region have been planted with multiple market classes obtained from different seed sources, including yellow, great northern, pinto, cranberry, black, navy, kidney, small red, and pink. Therefore the new epidemics could not be assigned to any particular instance or source. Disease incidence within fields has varied from trace levels to more than 90%.
 |
|
 |
|
Fig. 3. Leaf wilting symptoms and death of dry beans due to bacterial bean wilt. |
|
Fig. 4. Interveinal necrotic symptoms with irregular yellow haloes, characteristic of bean wilt. These symptoms are also referred to as "firing." |
First Report of a New Bacterial Disease
Bacterial wilt was first recognized and described as a new disease in 1922 near Redfield, SD on the farm of the Office of Forage Crop Investigations after the 1921 season. The grower on whose farm the disease was discovered informed the investigators that he believed the disease to be the same one that killed 90% of his crop in 1920. For the 1921 season, he planted the seed harvested from the previous year, resulting in an estimated 25% loss.
Florence Hedges
Florence Hedges was an associate pathologist with the USDA’s Bureau of Plant Industry (BPI), and was assigned the task by Erwin F. Smith to investigate this new disease outbreak. She was a native of Lansing, MI and worked most of her career for the BPI (Fig. 5). She began working on bean diseases for Smith after graduating from the University of Michigan in 1900, and was one of more than 20 women who worked in Smith’s lab before his death in 1927 (Fig. 6).
Smith was a rare progressive individual at that time who provided opportunities for women to work in the sciences in his lab (Fig. 7). The BPI, under Smith’s supervision, was the only federal agency to so willingly employ females as research assistants prior to World War I − a track record for which he was justifiably proud.
| |
 |
|
| |
Fig. 5. Florence Hedges (1878-1956).
(Photo credit: Smithsonian Institution). |
|
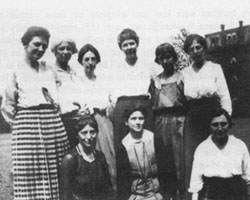 |
|
 |
|
Fig. 6. Women scientists working in the lab of E. F. Smith in 1921. From left to right: (standing) Charlotte Elliott, Hellie A. Brown, Edith Cash, Mary Katharine Bryan, Anna Jenkins, and Lucia McCulloch; (seated) Florence Hedges, Pearle Smith, and Angie Beckwith. (Photo credit: National Archives and Records Administration, Washington DC). |
|
Fig. 7. Laboratory of Plant Pathology, USDA, January 1905. Dean Smingle, Agnes Quirk, Florence Hedges, and Alice Haskins working in the lab. (Photo credit: National Archives and Records Administration, Washington DC). |
Investigating Bacterial Wilt
As one of the first efforts to begin addressing the problem, Hedges supervised the planting of a portion of the devastated 1920 South Dakota crop in Arlington, VA, resulting in a large number of diseased plants, many of which never survived beyond the seedling stage. After examining plants from both South Dakota and the experimental planting in Virginia she discovered yellow Gram-positive bacteria consistently colonizing the vascular system from wilted, diseased plants. These findings effectively ruled out the expected Gram-negative bacterial wilt organism Bacterium solanacearum, which had recently been discovered and characterized by Smith (now known as Ralstonia solanacearum).
After the initial finding in South Dakota, Hedges isolated the same pathogen from wilted navy bean plants grown in Michigan, Virginia, and Maryland, and from seed additionally collected from Montana, Michigan, Maryland, the District of Columbia, France, and Germany. She mentioned that based on this preliminary work in the early 1920s, she hypothesized that the disease appeared to be widely distributed throughout bean growing areas but had perhaps been previously confused with the more familiar bacterial disease known as common blight. She also opined that, with a high probability, the seedling wilt problems reported by the Plant Disease Survey, conducted from Indiana, Minnesota, New Jersey, Maryland, and Connecticut between 1917 and 1920 were also due to this pathogen. It was furthermore ascertained that as much as 90% of the crop in numerous Michigan fields in 1922 was attributed to this disease, making it the most damaging disease affecting beans in Michigan that season.
Koch’s postulates were fulfilled after repeated artificial inoculations with isolates from both the newly infected 1921 field-grown beans from South Dakota, and from the infected beans planted in Arlington, VA that were originally obtained from the 1920 crop from the same farm in Redmond, SD. The isolated organism from both sites was noted to be different from all other previously known pathogens, and named Bacterium flaccumfaciens (later transferred to Corynebacterium).
Pathogen Taxonomy and Terminology
The genus Corynebacterium was created in 1896 by K. B. Lehman and R. O. Neuman to house the diphtheria bacterium, C. diphtheriae. Soon after this other similar animal pathogens were placed into this genus followed by the addition of an increasingly wide array of superficially similar bacteria, making this genus a confusing repository for a catch-all group of Gram-positive, pleomorphic bacteria and other possibly related species.
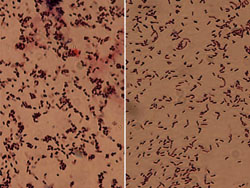
Hans Jensen, first noticed the similarity of certain soil coryneform bacteria and two specific plant pathogens, including those causing alfalfa wilt and tomato canker. He then suggested that these two phytobacteria be transferred to the genus Corynebacterium, reclassified them as C. insidiosum and C. michiganense, for the alfalfa and tomato pathogens respectively and created the term "coryneform" to refer to members of this group in general. This term today is still used to informally describe members of a heterogeneous group of Grampositive bacteria with cell morphological features consisting of irregular, short rods that may be slightly curved, bent, or club-shaped (Fig. 8), giving the genus its name from the Greek noun "koruna" or "club."
| |
 |
|
| |
Fig. 8. Comparison of morphological characteristics of two Coryneform bacterial pathogens. The shorter, fatter rods of Curtobacterium are on the left and the longer thinner rods of Clavibacter are on the right. |
|
In general, members of the plant pathogenic genus Corynebacterium were observed to cause two basic types of diseases: vascular wilts and hypertrophic diseases. The wilt group was further sub-divided into two groups based on other differing characteristics. One group included the previously mentioned C. insidiosum, C. michiganense, and the potato ring rot pathogen, C. sepedonicum, as slow growing, non-motile bacteria incapable of growth at temperatures above 35°C. The other group contained C. flaccumfaciens, C. betae, and C. poinsettiae as bacteria that grew more rapidly on nutrient media, had polar flagella and grew at 35-37°C.
The first three pathogens listed above were eventually transferred to the genus Clavibacter while the latter three are now pathovars of Curtobacterium flaccumfaciens, reflecting a group of closely related organisms that differ primarily in host range. The three pathovars flaccumfaciens, betae, and poinsettia of C. flaccumfaciens were then transferred into the new genus Curtobacterium on the basis of similar biochemical and DNA characteristics and cell wall composition. This new genus also was named etymologically based on its characteristic particle shape. The name Curtobacterium is derived from the Latin word "cutus" or "shortened" to describe a short bacterium or rod. The bean wilt pathogen originally obtained its species name from Florence Hedges after her seminal investigations to describe its most distinguishing characteristic. The specific epithet, flaccumfaciens is derived from the Latin word "flaccus" meaning "flabby or flaccid," and "faciens" meaning "making," thus describing the pathogenic actions of the bacterium and resulting symptoms as "wilt-making."
Hedges and Halo Blight
Florence Hedges’ landmark bacterial explorations were not limited to the wilt disease of beans. Her work with halo blight and kudzu is relatively obscure and should be better recognized today for its significant role in improving knowledge of bacterial diseases. Halo blight is another, distinct bacterial disease of dry beans caused by Pseudomonas syringae pv. phaseolicola. This disease first appeared in New York State in the early 1920s, and became a serious problem very rapidly. The demonstrated transmission of the pathogen through seed is presumed to explain its wide and rapid distribution. Pathogenicity of the organism was proven by Walter Burkholder who also determined it to be distinct from the previously identified common blight pathogen.
Common blight is caused by yet another bacterial pathogen, Xanthomonas axonopodis pv. phaseoli, and was recognized for the first time in the United States in 1892. Amazingly, the pathogen was demonstrated to be internally seedborne at this time before it was actually identified as Bacillus phaseoli in 1897 by E. F. Smith.
Simultaneously, during the mid-1920s when halo blight was being studied by Burkholder in New York, another disease of the leguminous vining plant, kudzu (Pueraria lobata) was being investigated independently by Hedges. In 1927, she isolated, described and named the causal agent Bacterium puerariae, but later determined that the kudzu disease was incited by the same pathogen that caused halo blight of beans. Because Burkholder’s description was published first, B. puerariae became a synonym, and the pathogen was later transferred to the genus Pseudomonas, where it resides today.
Nevertheless, based on Hedges’ stellar researches, it was brought to light that the bacterium was likely imported into the United States on the kudzu vine. This now highly noxious plant is thought to have been originally introduced from Japan into the United States in 1876 at the Philadelphia Centennial Exposition where it was used to cover the Japanese tea houses. A few plants were left behind for several inquisitive gardeners. Eventually the plant became of some economic importance as a forage crop and as a means for reducing soil erosion in the South.
However, this plant is amazingly invasive, capable of growing up to a foot per day during warm wet summers with more than 20 stems (runners) emerging from a single crown. Each of these runners is capable of reaching 100 feet in length with a single taproot weighing up to 400 pounds. Kudzu can endure severe winters, surviving in the ground as rhizomes, and is currently estimated to cover more than 7 million acres in the United States, with the further demonstrated ability to kill trees (Fig. 9) and pull down power lines. Not only is the plant now hugely problematic as a pervasive and meddlesome weed pest throughout the southeastern United States, but it also is now presumed to have served as the vehicle for unknowingly introducing a new pathogen into the United States that afterward became an important disease of beans.
| |
 |
|
| |
Fig. 9. Kudzu growth covering trees in North Carolina. |
|
Distribution of Bacterial Wilt
The exact distribution of the wilt pathogen today is uncertain, but it has been identified from Canada, Australia, Mexico, Brazil, and Tunisia. Although it has been additionally reported from numerous European Union countries including Belgium, Bulgaria, Greece, Romania, Russia, Hungary, Poland, Turkey, and Ukraine, it did not become established and is now considered to be absent from these countries. Apart from the pathogen being found in a few fields in southeastern Spain in 2005, no other recent records have been acknowledged from the EPPO region.
Within the United States, after the initial reports previously mentioned above, it continued to be detected sporadically, but widely distributed throughout the high plains and western U.S. bean-growing states between the late 1930s and the early 1950s. It was first noted in Wyoming in 1937, causing an estimated 1-2% damage, and a survey conducted 12 years later found the bacterium occurring by itself in 35% of fields and in combination with the common blight pathogen in another 28% of surveyed fields. In 1947, it was first observed from Idaho and from Colorado pinto fields but was not considered to be problematic at that time. The disease was further observed in 1948 from Nebraska and the Columbia Basin of Washington in 1951.
Hedges provided the first evidence of seedborne infection and transmission with the bean wilt organism, which was additionally reinforced as the disease was being newly discovered throughout the western United States. The first report from Idaho in 1947 was noted from several snap bean seed fields where severe infection and yield losses occurred, all of which originated from the same seed lot grown the year before, but presumably overlooked. That same year in Wyoming, severe losses exceeding 60% were documented in a field planted to beans the previous year while trace levels were observed in several other fields previously planted to other crops. Although the source of infection was never established conclusively, all fields in question utilized certified seed produced in Idaho, indicating, as Hedges postulated 20 years earlier, that infected seed served as the mechanism for pathogen infestation and dissemination into new areas.
New Pathogen Color Variants in Nebraska
The systemic nature of this disease was empirically demonstrated by Hedges and then formally proven by W. J. Zaumeyer later with electron microscopy. This trait enables the pathogen to move through the vascular (xylem) system into developing pods and seed embryos, often discoloring or staining seeds within pods (Fig. 10). This is particularly conspicuous on white seeded cultivars such as navy or great northern beans, which led directly to the discovery of different colored strains (often referred to as color variants) of the pathogen first in Nebraska (Fig. 11).
 |
|
 |
|
Fig. 10. A pod from a wilt-infected dry bean plant showing orange-stained seeds within pod. |
|
Fig. 11. Great northern dry bean seeds stained (infected) by pathogen color variants (reading clockwise from bottom) orange, purple, yellow, and uninfected. |
The initial report of bean wilt by Hedges from the eastern and mid-western United States described a bacterium with colony growth in culture and staining of infected seed that was always colored yellow (Fig. 11, top). Since the early 1950s, two additional distinctly colored Cff strains have been continually observed in Nebraska that were apparently restricted exclusively to bean fields in the western half of the state until the mid-1990s (Fig. 11, left and bottom).
Orange Strains
Bean wilt was first identified in Nebraska in 1948, and only four years later, orange-stained seeds (Fig. 12), yielding a bacterium producing orange growth in culture, were noted from 30% of cull samples collected from processing plants after harvest as part of a bacterial disease survey conducted during the 1952 season. Morphologically, physiologically, and serologically, the orange strains could not be distinguished from the original yellow strains other than by colony color, and this trait remained stable through multiple transfers and inoculations. However it was additionally observed that the tested orange strains were much more virulent than the original yellow strains. For example, studies screening varieties for resistance showed that pathogen populations detected within plants after inoculation were 5-10 times higher with the orange variants compared to the yellow and purple strains (see below) using three different bean varieties.
| |
 |
|
| |
Fig. 12. Dry bean plant surviving to harvest, but producing orange-colored, shriveled seeds as a result of wilt infection, compared to unaffected seeds from a different pod from the same plant. |
|
M. L. Schuster stated that orange-colored bacteria causing serious disease problems in field crops was a rare event, thus the appearance of an orange, highly virulent pathogen of dry beans in Nebraska was also assumed to be an unusual incident. However, over the next three years, the incidence of orange strains, documented by the presence of orange-stained seeds, increased to 8, 16, and 35% of the total infected seeds collected from 1953, 1954, and 1955, respectively. Schuster interpreted these data to mean that the differences that did exist between the orange and the original yellow strains were probably varietal and not specific. Thus he designated this "strain" as C. flaccumfaciens var. auranticum (orange-colored).
Purple Strains
It was not until 1967 that another Cff color variant was formerly identified and reported from western Nebraska. Great northern beans staining a purple to blue color were found within cull bean samples after processing. Growers and processors remarked that this phenomenon had occasionally been noted since the late 1950s, but had not been discovered during the initial 1952-1955 disease survey. By the late 1960s, the detectable incidence of this strain had increased to 2 seeds per pound (0.12-0.25%), and was named C. flaccumfaciens var. violaceae (violet-colored).
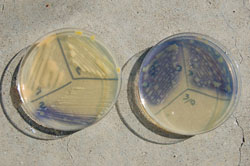
The purple strains were actually quite different from the orange variants by maintaining a yellow-colored colony in culture (not purple), but producing an extracellular, bright purple to blue, water-soluble pigment that diffused into the growth medium (Fig. 13) and also discolored seeds (Fig. 11, left). Pigment production varied by strains and was not correlated with culture age. Maximum pigment production was optimal at a temperature of 20°C and generally required 48 hours to be visualized, but some strains produced the pigments in as little as 15 hours. Fading and disappearance of pigments in culture tended to increase as temperatures increased, however the pigments in seed did not fade after 8 years storage at room temperature. The author has recently discovered that purple strains (at least those collected within the last decade in Nebraska) will more consistently produce the pigments if cultures are placed in the refrigerator for several hours or overnight after streaked bacterial growth appears on plates.
| |
 |
|
| |
Fig. 13. Bean wilt pathogen (purple color variant) growing in culture. Note the purple to blue pigments diffusing into media from well-defined yellow colored colonies. |
|
The purple variant has always been and still is very uncommon in occurrence compared to the yellow and orange strains. Other biological differences have also been seen with this unique organism. It was noted that the purple strains were slower growing, weaker, and less virulent than the other two variants, particularly the orange ones. Pigment production by the purple variant was often unstable and inconsistently expressed (as affected by temperature and pH) which may partially explain this variant’s lower reported incidence in nature. Without the pigment production, these isolates appear indistinguishable from the other yellow strains. Until 2006, this strain had never been reported outside of the Nebraska Panhandle, when it was detected from cull bean seeds in Alberta, Canada.
The purple and orange variants when originally found were considered to be of recent origin as they were not identified prior to 1948 (when wilt was first detected in Nebraska). Regardless of their obvious colorimetric differences, all three variants have a number of common characteristics such as similar DNA base composition and serological properties. Therefore, Schuster likely correctly considered the differences among the different strains to only warrant varietal status.
Pink Strains
As wilt re-emerged and incidence increased in the high plains region of the United States in the early to mid-2000s, all three pathogen color variants were discovered and isolated once again from infected dry bean plants originating from western Nebraska fields. More than 90% of collected isolates during this time consisted of the yellow and orange variants, further illustrating the lower incidence of the purple strains (6-8%).
Following the 2007 growing season, a pink bacterial isolate closely resembling the wilt pathogen was recovered from orange-stained seeds that originated from research plots affiliated with the University of Nebraska’s Panhandle Research and Extension Center (Scottsbluff Ag Lab) northwest of Scottsbluff, NE. The bacterium’s identity was confirmed as Cff based on several morphological and physiological characteristics. It reacted positively for both the Gram stain and KOH test, and exhibited short, pleomorphic rods characteristic of the coryneform bacteria (Fig. 8, left). Biolog identifications furthermore matched the pink isolate and Cff with a probability of 100 and 98% after 24 and 48 hours respectively. Pathogenicity tests revealed the pink strain to be highly virulent to dry beans, producing indistinguishable symptoms when compared to a standard known virulent orange isolate (Fig. 14).
| |
 |
|
| |
Fig. 14. Comparison of symptoms from plants inoculated with pink (left) and a standard highly virulent orange (right) isolate (15 days post-inoculation). |
|
The pink variant of bean wilt, like that of the orange strain, is very stable and appears to retain its colony color (Fig. 15) and virulence from the original isolation through multiple transfers and re-isolations from inoculated plants. Interestingly, this isolate was obtained from the same research farm that has yielded all three pathogen variants since 2005, and the same general area of western Nebraska from where the original orange and purple variants were first found. As a reprise, in 2011, another highly virulent pink variant was detected in association with a Goss’ wilt-infected corn plant (see below under "Alternate Hosts").
| |
 |
|
| |
Fig. 15. The pink wilt variant growing in culture compared to the typical yellow, orange, and purple variants on nutrient broth yeast extract medium (NBY). |
|
The presence of a fourth color variant for Cff in Nebraska, combined with the consistent occurrence of the other three pathogen variants for more than 50 years, further illustrates the remarkably high degree of microbial variation within this pathogen and among Nebraska dry bean production fields. We currently have no explanation for this, but Schuster postulated more than 35 years ago that this substantial pathogen divergence could potentially be due to the fact that Nebraska-produced seed is used principally for table food and not as seed beans, causing the distribution of new strains to be restricted to the bean growing areas of Nebraska where they presumably originated.
Conditions Favoring Disease
The wilt disease of beans is typical of many other bacterial diseases in that disease development is favored by a number of similar conditions. Important factors in general that favor the bacterial pathogens, promoting new infections and secondary spread throughout fields include planting infected seed, planting beans in close proximity to infected fields from the previous year, using reduced tillage methods of land preparation (Fig. 16), and environmental conditions involving high levels of leaf moisture.
| |
 |
|
| |
Fig. 16. Severely infested dry bean field in Nebraska planted into wheat stubble. This field was destroyed and never harvested due to extraordinarily high wilt incidence and severity. |
|
Furthermore, wilt is additionally enhanced by any factor that wounds bean plants such as storms, hail events, or any other physical damage from humans, animals, or farm equipment. Bacteria cannot readily infect plants by themselves, but instead require either natural openings in leaves, or wounding for initiating infection. Storms with high winds and hail provide the perfect opportunity for bacteria to become established because these events can cause wounding (Fig. 17) and can also physically move the pathogen and/or infected plant residues between and within fields. Burke first demonstrated experimentally that significant losses often occurred after hailstorms and was most extensive in fields where beans were grown regularly. He also found that infection still could occur in fields growing beans for the first time, even with certified disease-free seed.
| |
 |
|
| |
Fig. 17. Dry bean leaves damaged by hail. Note symptoms of bacterial infection associated with the holes in the tattered leaves. |
|
Disease development is also favored by high temperatures (> 30°C) with optimal pathogen growth in culture occurring at 27-28°C. Initial infection occurs when the pathogen enters the vascular system through either infected seed, the roots of developing seedlings, or through wounds on leaves or stems. However, bean wilt, in general, develops more rapidly and causes greater damage during periods of oppressive plant stress whether due to elevated heat levels, moisture deprivation, or mechanical damage of any kind.
Since the bean wilt bacterium is often soilborne as a result of residue colonization, Schuster conducted experiments to determine whether the wilt pathogen was spread through furrow irrigations. He determined that the bacterium was disseminated in the greenhouse but not from field studies, suggesting that spread in the field did not occur in this manner. R. M. Harveson and C. D. Yonts confirmed the same relationship of the pathogen with drip irrigation demonstrating that the pathogen did not move through soil to a great extent in the field, but it was demonstrated that the pathogen was capable of root to root spread in enclosed pot systems under greenhouse conditions.
Pathogen Survival
Longevity is important for the continued success of plant bacterial pathogens, therefore their survival depends on the ability to escape or endure adverse conditions. They do not form resting spores or other overwintering structures, remaining dormant during periods not conducive for infection like that of many fungi. Instead, bacteria depend upon other methods for survival until the occurrence of environmental conditions that favor infection.
The seedborne nature of Cff represents the major source of inoculum and means for dispersal, both long and short distance. However, it also provides an excellent mechanism for survival, both internally and superficially on the external seed surface (Fig. 18). In fact, the pathogen was demonstrated to survive and be re-isolated from infected seeds after being stored for 24 years in a glass jar held at room temperature on a laboratory shelf.
| |
 |
|
| |
Fig. 18. Wilt-infected, stained seeds in combine bin after harvest that serve as an overwintering source for the pathogen. |
|
As a rule of thumb, most coryneform bacterial plant pathogens do not survive well in soil without a close association with some form of crop residue, which is definitely the case for C. flaccumfaciens. Although the author determined in Nebraska that the wilt pathogen can survive in soil in the absence of residue for at least two years, resulting infection percentages of emerging seedlings after planting healthy seeds into these infested soils remained very low (<2%) (Fig. 19).
| |
 |
|
| |
Fig. 19. Dry bean seedlings emerging from pathogen-infested but residue-free soil in the greenhouse. Note the single wilted plant that became infected sometime during the germination and emergence process. |
|
Alternate Hosts
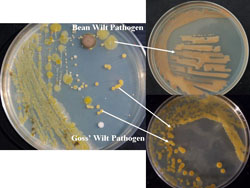
Over the last seven to eight years, dry bean-pathogenic bacterial isolates associated with other crops commonly grown in rotation with dry beans have been isolated and identified by the author after extensive surveys of Nebraska production fields. Approximately two dozen wilt-like isolates have been collected from soybean, corn, wheat, alfalfa (Fig. 20), and sunflower, further illustrating the widely distributed nature of the pathogen throughout western Nebraska production fields. All of these field crops are additionally susceptible to their own bacterial diseases, and these virulent, wilt-like isolates were always found in association with other recognized phytobabacterial diseases, such as Goss’ wilt of corn (Fig. 21) and black chaff of wheat, not only suggesting survival in these crop residues, but also as saprophytes in live, but bacterial-infected plants (Fig. 22).
| |
 |
|
| |
Fig. 20. Wilt symptoms on dry bean plants inoculated by isolates collected from other bacterial-infected crops grown in rotation with dry beans. Reading clockwise from upper left: isolates associated with soybean, corn, wheat, and alfalfa. |
|
 |
|
 |
|
Fig. 21. Bacterial colonies streaked on media from a Goss’ wilt-infected corn leaf that illustrate the methodology for separation and identification of multiple pathogens present in bacterial-infected leaves. |
|
Fig. 22. Examples from bacterial-infected alternate crops from which wilt strains pathogenic to dry beans were isolated. (Goss’ wilt of corn on left, and black chaff of wheat on right). |
The author has previously established relationships between dry beans and Cff isolates obtained from soybeans. Both soybean and dry bean isolates were found to be pathogenic on both crops after reciprocal inoculations were performed. However, all isolates were more virulent on dry beans than soybeans, regardless of host origin, suggesting that dry beans are more susceptible to the pathogen than soybeans. It is not known for certain how collected strains of this pathogen may affect their respective isolation hosts of corn, wheat, alfalfa, sunflower, or potentially others as yet untested, but at the very least, these additional crops apparently can still benefit the pathogen by serving as another source of survival as well as providing a source of inoculum for infecting dry beans when they are put back into the rotation.
Conclusions and Summary
The resurrection of this disease in Nebraska and other areas of the high plains after a long absence has been a puzzling development. Because of the historical seedborne nature of this problem, it raises the question of whether seed sanitation in areas of increase has been forgotten or ignored. When the disease reemerged in Nebraska late in the 2003 season, it was assumed to be an isolated incident. However, the next season the pathogen was found to be widespread throughout western Nebraska production fields, and has now been identified from well over 400 fields across this region since 2004.
It is unclear as to exactly why this disease has returned again at this time. Some possibilities include contaminated seed lots, cultivation of extremely susceptible cultivars, growth and survival in other hosts grown in rotation with beans, or changes in the pathogen or its population structure. For example, evidence has been obtained suggesting that strains collected in 2004-2008 appear to be more aggressive and virulent than historical reference strains that have been in storage for 30+ years.
Another factor in this re-emergence story may be explained conjecturally by the climatic patterns observed throughout the region over this same period. The mid 2000s were characterized by milder winters, and an extended drought with increasingly higher summer temperatures during the growing season. These factors could have easily contributed to better conditions for pathogen survival and increased plant stresses, which are well recognized to favor wilt disease development and increased severity.
Lastly, the changes in cultural practices that have been adopted in this region over the last 15-20 years have also played perhaps the most influential role in this mystery. The majority of area producers have introduced some form of reduced tillage into their systems. Combining this practice with the simultaneous, region-wide increases in center pivot sprinkler irrigation systems would reputably provide better conditions for enhancing pathogen survival, infection, and dispersal within fields.
In summary, after almost a decade of studying this disease, the author maintains that it has "reappeared" and become widely established throughout the dry bean production areas in this region, due in large part, to our recent changes in cultural practices, although other factors have undoubtedly also played a role. It seems likely that the pathogen never disappeared completely, but was able to survive at low levels as a saprophyte on weeds, other crop species, or plant residues. Thus it was not necessarily noticed until recently because in the past most fields were routinely plowed each season (removing a major source of survival). It is also logical to hypothesize that the disease was simply overlooked (or mis-identified) due to the fact that the symptoms of wilt are reminiscent of and potentially confused with common blight, a prescient observation first remarked by Florence Hedges almost 90 years ago. The recent data collected and presented here strongly suggest that the return of bean wilt throughout the Central High Plains over the last decade is not due to a single factor but a combination of new changes in cultural practices, environmental stresses, and unfamiliarity with the pathogen and its past history, creating a perfect storm of conditions that encouraged and enhanced new destructive epidemics.
References and Further Reading
Agarkova, I. V., Lambrecht, P. A., Vidaver, A. K., and Harveson, R. M. 2012. Genetic diversity of Curtobacterium flaccumfaciens pv. flaccumfaciens populations in the American High Plains. Can. J. Microbiol. 58:788-801.
Burke, D. W. 1957. Incidence of bacterial pathogens in dry beans in irrigated districts of Nebraska, Wyoming, and Colorado in 1954 and 1955. Plant Dis. Rep. 41:488-490.
Burke, D. W. 1957. Bacterial wilt of pinto beans on soils of different types and cropping histories. Plant Dis. Rep. 44:671-673.
Burke, D. W., and Seliskar, C. E. Disease incidence and yields of beans in relation to cultivation in northeastern Colorado. Plant Dis. Rep. 41:483-487.
Burkholder, W. H. 1926. A new bacterial disease of the bean. Phytopathology 16:915-927.
Burkholder, W. H. 1945. The longevity of the pathogen causing the wilt of common bean. Phytopathology 35:743-744.
Collins, M. D., and Jones, D. 1983. Reclassification of Corynebacterium flaccumfaciens, Corynebacterium betae, Corynebacterium oortii, and Corynebacterium poinsettiae in the genus Curtobacterium as Curtobacterium flaccumfaciens comb. nov. J. Gen. Microbiol. 129:3545-3548.
Davis, M. J. 1986. Tazonomy of plant-pathogenic coryneform bacteria. Ann. Rev. Phytopathol. 24:115-140.
Davis, M. J. 2001. Coryneform phytobacteria. Pages 255-256 in: Encyclopdia of Plant Pathology, Vol 1. O. C. Maloy and T. D. Murray, eds. John Wiley and Sons, Inc. New York, NY.
EPPO/CABI. 1997. Curtobacterium flaccumfaciens pv. flaccumfaciens. Pages 991-994 in: Quaratine Pests for Europe, 2nd Edn. I. M. Smith, D. G. McNamara, P. R. Scott, and H. Holderness, eds. CAB Intl., Wallingford, UK.
Harveson, R. M. 2009. New hosts for the dry bean bacterial wilt pathogen in western Nebraska? Phytopathology 99:52.
Harveson, R. M., Schwartz, H. F., Vidaver, A. K., Lambrecht, P. A., and Otto, K. 2006. New outbreaks of bacterial wilt of dry beans in Nebraska observed from field infections. Plant Dis. 90:681.
Harveson, R. M., and Schwartz, H. F. 2007. Bacterial diseases of dry edible beans in the Central High Plains. Online. Plant Health Progress doi:10.1094/PHP-2007-0125-01-DG.
Harveson, R. M., and Vidaver, A. K. 2007. First report of the natural occurrence of soybean bacterial wilt iolates pathogenic to dry beans in Nebraska. Online. Plant Health Progress doi:10.1094/PHP- 2007-0822-01-BR.
Harveson, R. M., and Vidaver, A. K. 2008. A new color variant of the dry bean bacterial wilt pathogen (Curtobacterium flaccumfaciens pv. flaccumfaciens) found in western Nebraska. Online. Plant Health Progress doi:10.1094/PHP-2008-0815-01- BR.
Harveson, R. M., and Vidaver, A. K. 2009. Alternate hosts for the dry bean bacterial wilt pathogen in western Nebraska? Annu. Rept. Bean Improv. Coop. Abst. P-30. Ft. Collins, CO.
Harveson, R. M., and Yonts, C. D. 2007. Influence of irrigation method on incidence and severity of bacterial wilt of dry beans in Nebraska. Phytopathology 97:S45.
Hedges, F. 1922. A bacterial wilt of the bean caused by Bacterium flaccumfaciens nov. sp. Science LV: 433-434.
Hedges, F. 1926. Bacterial wilt of beans (Bacterium flaccumfaciens Hedges), including comparisons with Bacterium phaseoli. Phytopathology 16:1-22.
Hedges, F. 1926. Bean wilt traceable to infected seed. Pages 165-166 in: Yearbook of Agriculture. USDA, Washington, DC.
Hedges, F. 1927. Bacterial halo spot of kudzu. Phytopathology 17:48.
Hedges, F. 1928. Bacterial halo spot of kudzu caused by Bacterium puerariae Hedges. J. Agr. Res. 36:419-428.
Hedges, F. 1930. The relationship of Bacterium medicaginis var. phaseolicola and Bacterium puerariae. Phytopathology 20:140.
Hsieh, T. F., Huang, H. C., Erickson, R. S., Yanke, L. J., and Mundel, H.-H. 2002. First report of bacterial wilt of common bean caused by Curtobacterium flaccumfaciens in western Canada. Plant Dis. 86:1275.
Huang, H. C., Erickson, R. S., Yanke, L. J., Chelle, C. D., and Mündel, H.-H. 2006. First report of the purple variant of Curtobacterium flaccumfaciens pv. flaccumfaciens, causal agent of bacterial wilt of bean, in Canada. Phytopathology 90:1262.
Lehman, K. B., and Neumann, R. O. 1896. Atlas und Grundriss der Bakteriologie und Lehrbuch der speziellen bakteriologishen Diagnostik. 2:1-448. 1st Edn. Lehman, Munchen.
Lelliott, R. A. 1966. The plant pathogenic coryneform bacteria. J. Appl. Bact. 29:114-118.
Jensen, H. L. 1934. Studies on saprophytic mycobacteria and corynebacteria. Proc. Linn. Soc. N.S.W. 59:19-61.
Jensen, H. L. 1952. The coryneform bacteria. Ann. Rev. Microbiol. 6:77-90.
Matta, C. 2008. Early women in the United States and at the U. S. Department of Agriculture: Charlotte Elliott. Pages 47-57 in: Pioneering Women in Plant Pathology. American Phytopathological, St. Paul, MN.
Menzies, J. D. 1954. Effect of sprinkler irrigation in an arid climate on the spread of bacterial diseases of beans. Phytopathology 44:553-556.
Ristaino, J. B. 2008. Introduction. Pages 9-11 in: Pioneering Women in Plant Pathology. American Phytopathological, St. Paul, MN.
Schuster, M. L. 1959. Relation of root-knot nematodes and irrigation water to the incidence and dissemination of bacterial wilt of bean. Plant Dis. Rep. 43:27-32.
Schuster, M. L., and Christiansen, D. W. 1957. An orange-colored strain of Corynebacterium flaccumfaciens causing bean wilt. Phytopathology 47:51-53.
Schuster, M. L., and Coyne, D. P. 1974. Survival mechanisms of phytopathogenic bacteria. Ann. Rev. Phytopathol. 12:199-221.
Schuster, M. L., and Coyne, D. P. 1975. Genetic variation in bean bacterial pathogens. Euphytica 24:143-147.
Schuster, M. L., and Coyne, D. P. 1975. Survival factors of plant pathogenic bacteria. Research Bulletin 268, University of Nebraska, Lincoln, NE.
Schuster, M. L., and Sayre, R. M. 1967. A coryneform bacterium induces purple-colored seed and leaf hypertrophy of Phaseolus vulgaris and other Leguminosae. Phytopathology 57:1064-1066.
Schuster, M. L., Vidaver, A. K., and Mandel, M. 1968. A purple pigment-producing bean wilt bacterium Corynebacterium flaccumfaciens var. violaceum n. var. Can. J. Microbiol. 14:423-427.
Stewart, A. 2009. Wicked plants: The weed that killed Lincoln’s mother and other botanical atrocities. Algonquin Books, Chapel Hill, NC.
Thomas, W. D., and Graham, R. W. 1952. Bacteria in apparently healthy pinto beans. Phytopathology 42:214.
Venette, J. R., Lamppa, R. S., and Gross, P. L. 1995. First report of bean bacterial wilt caused by Curtobacterium flaccumfaciens subsp. flaccumfaciens in North Dakota. Plant Dis. 79:966.
Vidaver, A. K., and Starr, M. P. 1981. Phytopathogenic coryneform and related bacteria. Pages 1879-1887 in: The Prokaryotes, A Handbook on Habitats, Isolation, and Identification of Bacteria. Springer-Verlag, New York, NY.
Vidaver, A. K. 1982. The plant pathogenic corynebacteria. Ann. Rev. Microbiol. 36:495-517.
Zaumeyer, W. J. 1932. Comparative pathological histology of three bacterial diseases of bean. J. Agric. Res. 44:605-632.
Zaumeyer, W. J. 1944. A monographic study of bean diseases and methods for their control. USDA Technical Bull. 868. USDA, Washington, DC.
Zaumeyer, W. J., and Thomas, H. R. 1947. Bean diseases in some of the mountain states in 1947. Plant Dis. Rep.31:432-442.
