Caitilyn Allen, Andrew Bent, and Amy Charkowski
Department of Plant Pathology,
University of Wisconsin-Madison,
Madison, WI 53706
(Corresponding author: cza@plantpath.wisc.edu)


Introduction
Despite rapid advances on certain aspects of plant pathogenic bacteria, many economically important pathosystems are largely unexplored and biologically relevant life stages of even familiar systems remain poorly understood. We know remarkably little about end-stage disease, latent infections, survival away from the host, interactions among multiple microbes in a plant, and the effects of quantitative virulence factors. While no thoughtful researcher would dispute the effectiveness of reductionist experiments, we propose that this approach be combined with a broader perspective that includes the ecology, histopathology, and community population biology of phytopathogenic bacteria. We offer examples of exciting recent discoveries resulting from this natural history-based approach. In particular, in situ studies using biologically realistic inoculation followed by analyses with microscopy, gene expression profiling, community analyses, or application of key computational tools can offer new insights into old questions. Research that combines cutting-edge tools with a biological perspective is especially lacking on high-impact diseases of subsistence crops. Understanding the biology underlying important practical issues such as copper resistance, eradication from seed and cuttings, and rapid, sensitive detection could be of significant utility. Overall, we endorse a broader biological approach to research on plant pathogenic bacteria.
Choices, Choices
In response to significant advances in plant bacteriology, researchers can focus in to more deeply understand the discovery, or they can change the subject and turn to important questions that remain poorly understood. This article encourages the second approach by pointing out some underexplored but important aspects of plant pathogenic bacteria. We first discuss considerations that may aid selection of research topics, and then suggest a necessarily incomplete set of specific questions and approaches that promise fresh and productive research.
Ideally, our research programs would be designed to reveal fundamental biology of high-impact plant pathogens, leading to useful disease management strategies. All too often our planning instead brings us to the intersection of the feasible, the fundable, and the familiar — hardly a path to novelty. We suggest that those seeking new directions should instead choose a study system that satisfies at least two of the criteria listed in Table 1. In particular, research is urgently needed on destructive diseases of key tropical subsistence crops, such as Xanthomonas wilt of banana (Musa spp.) and bacterial blight of cassava (Manihot esculenta). A more widespread focus on research to reduce crop losses offers the additional benefit of increasing stakeholder support for plant bacteriology funding.
Table 1. Some criteria to identify novel and important systems for research.
| Major disease of major staple crop |
Disease of understudied staple crop (e.g. plantains, oil palms,
cassava) |
| Major disease of high-value specialty crop or developing nation crop |
| Effective disease management would expand cropping zone |
Commodity group or international non-government organization
support |
| Current control methods environmentally undesirable |
| Pathogen persistence in environment |
| Pathogen colonization of plant surface or vasculature |
| Pathogen latent or commensal stage |
| Pathogen seed transmissibility |
| Pathogen insect transmissibility |
| System has unique biology (e.g. Agrobacterium tumefaciens) |
| Plant–associated human pathogen |
| Pathosystem has potential impact on medical biology
|
Look Back to Move Forward
New knowledge and methods create opportunities for progress on old questions, and indeed there are few truly new questions. It is humbling to discover that our scientific predecessors thought deeply and usefully about our subject. Perceptive articles and book chapters that were written long before the advent of PubMed can be overlooked in an online search. Readers curious about plant pathogenic bacteria are encouraged to explore the following and other older sources, which describe key research questions that remain unsolved (Smith, 1920; Walker, 1963; Schuster and Coyne, 1974; Vidaver, 1981; Mount and Lacy, 1982; Starr, 1984; Billing, 1987; Nester et al., 2004). In the same spirit, readers are encouraged to remain open to the curiosity about the natural world that drew us to science. Fancy tools are one route to novel findings, but paradigm-shifting discoveries often come from simple observation. Charles Darwin had travel funds, notebooks, pencils, and a few dead birds.
The Pendulum is Swinging Toward Diversity
The early years of molecular plant bacteriology explored a wide range of interactions. Conducted without kits, PCR, or commercial DNA sequencing, this research used laborious methods such as screening and characterization of transposon mutants to discover hrp (for host response and pathogenicity) and avr (for avirulence) genes in the interactions between Pseudomonads and bean (Phaseolus vulgaris) plants, dissect the role of cell wall-degrading enzymes in soft-rot enterobacteria, and determine that EPS is key to wilt pathogenesis (Staskawicz et al., 1984; Niepold et al., 1985; Lindgren et al., 1986; Kotoujansky, 1987; Denny and Baek, 1991). The rapid discovery of the unique mechanisms underlying crown gall disease demonstrated how quickly an area could advance given significant investment and competition (Zambryski, 1988). This insight, together with the rise of Arabidopsis (Arabidopsis thaliana) as a host and enthusiasm for model systems in general, has drawn molecular plant bacteriologists to a narrow set of pathosystems. Grant proposals often argue that results obtained with these organisms will be easily applied to economically important plant diseases. However, the pioneering bacterial geneticist Jacques Monod was famously wrong when he said that “anything found to be true of E. coli must also be true of Elephants” (Monod and Jacob, 1961, p. 393). While many mechanisms are common across biological systems, even closely related organisms have adapted and shaped ancestral tools to diverse ends, solving similar problems in strikingly different ways or adapting the same protein for unrelated functions. On the heels of success with model systems, a renewed effort to study diverse plant-bacterial systems is needed precisely because what is true for DC3000 in an Arabidopsis leaf is not always true for Xylella fastidiosa in a grapevine (Vitis spp.). Understanding gene-for-gene resistance to bacterial blight of rice (Oryza sativa) is important, but it is unlikely to elucidate horizontal resistance to bacterial wilt in tomato (Solanum lycopersicum) or lead to greening-tolerant citrus trees (Citrus spp.).
Bacterial Behavior in Natural Hosts Under Biologically Realistic Conditions is Under-Explored
Reductionist experiments are powerful, but the lure of their yes/no results can keep us from doing discovery experiments that may be complicated and messy but also more biologically realistic and practically relevant. Familiar examples include studies focusing on single genes rather than multigenic traits, model systems instead of natural hosts, sterile potting mix in place of natural soil, seedlings rather than mature plants, and controlled rather than field environments. Every researcher struggles to balance experimental feasibility with biological meaning, but a convenient and familiar assay can give deceptive results that hide a more interesting truth. For example, He and co-workers found that the phytotoxin coronatine facilitates pathogen entry into leaf mesophyll by causing stomates to open, but this effect was masked if leaves were infiltrated with bacteria and was only detectable when Pseudomonas syringae strains were inoculated onto leaf surfaces (Melotto et al., 2006). Similarly, a series of epidemiological studies of P. syringae as a bean epiphyte and pathogen by Hirano and Upper laid the foundation for elegant experiments showing that Type III secreted effectors and the Gac regulon are each critical for epiphytic fitness in the field; these important phenotypes were invisible in the controlled environment of a growth chamber (Upper and Hirano, 1996; Hirano et al., 1997; Hirano et al., 1999). Seedlings of the apple (Malus domestica) rootstock Budagovsky 9 appear to be susceptible to fireblight, but the mature woody trees are disease-resistant in the field (Russo et al., 2008).
What Are They Up To Behind Our Backs?
Pathologists have traditionally, and understandably, focused on discovering how bacteria incite disease during the early stages of acute pathogenesis. We know much less about the end stages of bacterial pathogenesis, how bacteria escape from dying plants, and the traits needed to grow or persist in free-living states in soil, drainage ditches, dead plant residues, on farm implements, or up in the sky. Some species must colonize seeds, vectors, or alternate hosts; others form lesions or other structures that foster bacterial spread in the environment. Although most plant pathogenic bacteria do not form spores, they often survive extremes of humidity and persist for years; how? There are fascinating biological questions in these understudied life stages, which can be found in the disease cycle of almost every plant pathogenic bacterium. Figure 1 provides only one of many illustrative examples. There are significant opportunities for improved disease control if any stage of the disease cycle can be disrupted.
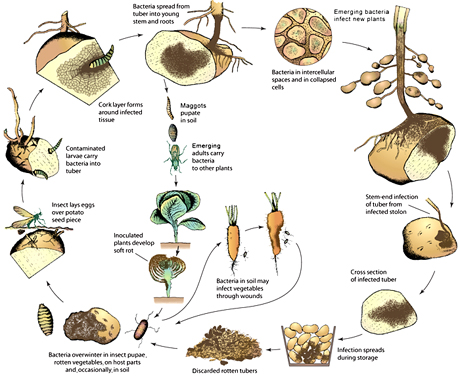
Fig. 1. An example of the multiple relevant life stages in the disease cycle of plant pathogenic bacteria: Soft-rot of vegetables caused by pectinolytic enterobacteria in the genera Erwinia, Pectobacterium, and Dickeya. Reprinted with permission from Plant Pathology (5th edition) 2005, by George Agrios, Elsevier Academic Press, Burlington, MA.
Less-Examined life Stages Are Now More Accessible
Detection methods such as real-time PCR, GFP tags, and immunofluorescence staining microscopy are sensitive enough to study small populations in situ in the rhizosphere, in water, in animals, and on soil particles. We do not know much about associations between plant pathogenic bacteria and native plants, especially the roles of these bacteria in natural ecosystems. The extent of our ignorance is exemplified by the recent discovery that the very well-studied soft-rot bacterium Erwinia chrysanthemi (now Dickeya dadantii) has a secret life as an insect pathogen (Grenier et al., 2006).
Accumulating evidence suggests the ubiquity in plants of bacterial endophytes, most of which are currently unculturable (Zinniel et al., 2002). These largely unexplored communities likely affect disease development (Araújo et al., 2002) and endophytes are also potential biocontrol agents or delivery systems for antipathogenic compounds (Kobayashi and Palumbo, 2000). Further, important plant pathogens like Ralstonia solanacearum, Liberibacter asiaticus, X. fastidiosa, and Clavibacter sepidonicum cause long-term latent infections, effectively functioning as endophytes. What biological signals or conditions tip the balance and cause an innocuous endophyte to become a destructive pathogen? Metagenomic community analysis and in situ transcriptional studies can open windows into this previously inaccessible aspect of plant microbiology.
The discovery that some plant pathogenic bacteria affect the weather when they are not living on plants (Christner et al., 2008; Morris et al., 2008) further demonstrates the importance of looking beyond the acute pathogenesis stage of the disease cycle and beyond crop hosts, as well as the power of crossdisciplinary collaborations. However, moving out of the one-gene/one-trait, plus-or-minus-assay comfort zone demands transdisciplinary approaches. For example, collaborations with epidemiologists who have expertise in the relevant statistical and modeling tools can reveal the mechanisms of subtle but crucial quantitative traits like competition, survival, and dispersal.
Getting Leverage on Quantitative Problems and Systems Biology
Biological interactions are dynamic, with balances sometimes tipping sharply when thresholds in signaling or population levels are reached. Environmental variability (humidity, temperature, drought stress) has significant but largely unknown effects on plantbacterial interactions. Metagenomics have shown us that rhizospheres and leaf surfaces support complex communities of microbes that are mostly uncultured and undescribed (Riesenfeld et al., 2004). These communities almost certainly affect the behavior of plant pathogens, but most experiments ignore them. However, we now have computational tools and extensive genomic and gene expression data that allow us to model complex traits of interest to both phytobacteriologists and mathematicians. These instruments can add quantitative information for regulatory models; elucidate complex phenomena like the initiation of infection (Sepulchre et al., 2006) or bacterial cell differentiation (Craciun et al., 2006); identify emergent properties in bacteria and host plants (Long et al., 2008); and model the complex microbial communities that are important for both pathogenesis and biocontrol (Gilbert et al., 1993, 1996; Schloss and Handelsman, 2008). Many of the quantitative methods needed to design and analyze these kinds of experiments are familiar to ecologists and statisticians, who thus make excellent collaborators for molecular bacteriologists.
The Underexploited Power of the Microscope
Natural history remains a powerful form of biology, as everyone who is annotating genomes can testify. It is becoming clear that genomics and even gene expression studies cannot deliver the specific insights offered by using microscopy to follow bacterial colonization and pathogenesis in real time. High-quality histopathology is time consuming and technically demanding, but it is highly rewarding to use superior modern instruments to observe specifically labeled cells, structures, or proteins in situ. This approach has been very productive in studies of animal pathogenesis but is underused by plant microbiologists (but see Newman et al., 2003; Meng et al., 2005; Monier and Lindow, 2005; Melotto et al., 2006; Nakaho and Allen, 2009). Microscopy studies often suggest hypothesis-driven experiments using defined mutants, and this combination can produce especially rapid advances, such as the fascinating discovery that X. fastidiosa uses twitching motility to move against the transpirational flow in xylem and colonize below an infection point (Meng et al., 2005). Similarly, microscopy and fluorescent probes were combined with genetics to gauge the distance signal molecules can travel between cells on roots and leaves (Gantner et al., 2006; Dulla and Lindow, 2008).
Hypothetically Speaking
Annotation reinforces the conventional wisdom because we can confidently identify only those genes that have been previously studied. The rapidly expanding set of genomes for plant pathogenic bacteria can be combined with powerful bioinformatics tools and a biologist’s perspective to generate some fresh hypotheses about the many conserved hypothetical proteins crowding our genome databases. For example, straightforward experiments would be suggested by the discovery that a particular conserved gene of unknown function is present in genomes of all insect-transmitted bacteria, no matter how distantly related, but absent from genomes of closely related species transmitted by other means. Similar analyses can find conserved hypothetical proteins specific to epiphytes, xylem dwellers, bacteria attacking only monocots, etc. An analysis of plant pathogenic Xanthomonas genomes used this idea to identify genes potentially linked to infecting specific tissues (Lu et al., 2008). However, the next steps require a better understanding of the hidden lives of bacterial pathogens than we currently have, highlighting the need for a greater overlap with bioinformatics and natural history. A word of caution as we discuss bioinformatics and annotation: it is important to test the biological function of a putative gene before drawing too many conclusions. It would be unfortunate and ironic if, in our excitement about genomics, we allowed mutant construction and phenotypic testing to become an underexplored research niche.
Other Underexploited Methods
Microarray-based profiling of pathogen gene expression under various conditions is advancing at a rapid pace, although more expression studies are needed in biologically relevant in planta settings. Gene expression profiling can be coupled with laser capture microdissection or other creative extraction methods so that specific microbial subpopulations (or specific host cells or tissues) can be analyzed with increased sensitivity. In vivo expression technology screens and their offshoots (Osbourn et al., 1987; Rainey and Preston, 2000; Boch et al., 2002; Brown and Allen, 2004) have still not been employed to detect genes that are expressed at key growth stages of many important plant pathogens. Biosensors offer a way to measure specific conditions as experienced by bacteria in planta (Wright and Beattie, 2004). Semirobotic sample processing, ordered gene knockout collections, and heavy isotope labeling are other examples of promising methods. These are only a few of the underused technologies; the broader microbial sciences offer a regular flow of new methods that beg for use in the study of plant pathogens. As noted above, an equally important source of new methods is collaboration with experts from other disciplines. Partnership with the right ecologist, computer scientist, microfluidics specialist, geologist, or others can break open a previously recalcitrant problem. Finally, getting out into the field to see pathogens under natural conditions often suggests fresh experimental methods or questions.
Multifunctional (Cross-Kingdom) Signaling
Many of the same signal molecules are perceived by both plants and microbes. This is not surprising since angiosperms arose about 3 billion years after bacteria, and evolved in the constant presence of microbial signaling. Similar signal molecules are produced by a wide range of bacteria and all bacterial plant pathogens are likely to be exposed to plant signal molecules, yet the roles of these signals on plants and bacteria has only been explored in a handful of pathosystems and even fewer have been placed into signal networks (Brencic and Winans, 2005). In these cases, it is clear that bacteria are integrating signals from both plant and bacterial cells to regulate virulence genes at the transcriptional and posttranscriptional level, although the relative strength and timing of each signal remains obscure. For example, soft-rot pathogens use a combination of auxin, pectin metabolites, acyl-homoserine lactones, and organic acids to regulate pectate lyases and other virulence genes at both transcriptional and posttranscriptional stages and it is possible to interfere with soft-rot pathogenicity by disrupting these signaling cascades (Charkowski, 2009). Similarly, Agrobacterium responds to auxin, g-amino butyric acid, and salicylic acid (Yuan et al., 2008). It is also clear that signaling cascades are modular since closely related bacteria use identical signal molecules and receptors in different ways. Therefore, these cascades need to be examined in multiple species to understand how plants and bacteria are manipulating each other with small molecules.
Aspects of small molecule signaling and defense studied in other areas of microbiology have not yet made large impacts on plant pathology. In some cases, signaling properties of classes of molecules, such as flavonoids, have been described in detail by those examining beneficial microbes such as Rhizobium (Gibson et al., 2008), but we have barely scratched the surface in determining if and how the same class of compounds affect signaling in pathogens. As an example, flavonoid glycosides induce SyrB, which is required for synthesis of syringomycin by P. syringae (Mo et al., 1995). Similarly, many plant compounds have been examined for their antimicrobial effects on human pathogens, but their effects on plant pathogens are unknown. For example, 5’-methoxyhydnocarpin, a plant compound that inhibits an ATP-binding cassette transporter, thereby making the plant-produced antibiotic berberine more effective, has been studied for its effect on a human pathogen, but not on plant pathogens (Stermitz et al., 2000).
Identifying Hidden Partnerships
Plants face multiple pathogens and there are hints that some pathogens function best in pairs, but this area has been little explored. An almost completely unexamined example is soft-rot disease caused by Clostridium. Clostridium and Pectobacterium species are routinely found together in decaying vegetables and both can cause disease on their own (Pérombelon et al., 1979; Campos et al., 1982). These pathogens may work together to attack their plant hosts. Although potatoes (Solanum tuberosum) are mostly starch, Pectobacterium curiously cannot degrade starch, while Clostridium efficiently breaks down this polymer. Close relatives of Pectobacterium, such as Klebsiella, can metabolize starch (Holt, 1994), so the inability to use this abundant polymer is not inherent, but perhaps developed under the selection of the Pectobacterium-Clostridium partnership. This partnership, as well the role anaerobes play in the soil and on roots and the effects of low oxygen on bacterial pathogens in general, is little explored. This may be because plant pathologists are reluctant to work with anaerobes. Nevertheless, pathogens face low-oxygen environments in plants, in soil, and in waterways, suggesting that research on this topic could be fruitful.
Microbe-Associated Molecular Patterns and Effectors
Two areas of phytobacteriology that are currently under intensive study are microbe-associated molecular patterns (MAMPs; also called PAMPs) and type III secretion system-dependent effector proteins. Our primary message is to encourage research beyond these heavily studied topics, but even these topics contain underexplored niches concerning the real-world relevance of MAMPs and effectors (Bent and Mackey, 2007). For example, in the biologically realistic setting of an intact plant infested with a reasonable population of living microorganisms, how much MAMP is present and needs to be present, and in what plant tissues, for effective defenses to be triggered? Are epidermal cells, which are routinely exposed to an extensive microbial flora, less sensitive to MAMPs? Learning how plant cell types differ in their responses would help us determine how MAMP detection systems work along natural routes of bacterial entry and spread, such as the vasculature. This could also move us toward understanding how many host processes need to be inhibited by effectors for any particular pathogen to succeed, and if different pathogen species commonly suppress the same host targets. The role of effectors in gene-for-gene systems is well studied, but are defense-suppressing effectors important for broad host-range pathogens as well? Why are plant resistance genes that work against necrotrophs so rare? It has been suggested that necrotrophs are less dependent on suppression of plant defenses and may even benefit from induction of some defense pathways (Glazebrook, 2005), but this hypothesis remains to be broadly tested. Similar unanswered questions remain about the real-world biology, especially in field settings, of other much-studied virulence factors like toxins, plant growth regulators, and macerating enzymes.
Several major problems in the management of bacterial plant diseases could be solved with a better understanding of the underlying biology. A few examples are given below.
Copper and Antibiotic Resistance
Some core methods for control of bacterial diseases, such as copper or streptomycin sprays, lose their utility because pathogens become resistant, often through acquisition of broad host-range transmissible plasmids from other bacteria. Can anything be done to prevent this? Alternatively, β-lactamase inhibitors like clavulanic acid are used clinically to make amoxicillin work against resistant strains (Payne et al., 1994); can this approach be adapted to agricultural ecosystems? Are there copper resistance protein inhibitors in the soil metagenome?
Eradication from Seeds, Cuttings, and Seedlings
Seed treatments such as hot water treatments are one of the best interventions available to disrupt bacterial diseases, but they are only effective in some pathosystems (Leben and Sleesman, 1981). Why? Is it simply a pragmatic issue of accessing vulnerable bacteria without killing the seeds, or are there more interesting aspects of bacterial biology that underpin resistance to such treatments? What controls the ability of bacteria to colonize certain regions of seed or sanctuaries in other tissues, or the ability of plant seeds to tolerate antibacterial treatments?
Pathogen Detection
Research seems to have dwindled on the previous two problems, but ongoing efforts seek improved detection methods for bacteria on seeds and cuttings (Gitaitis and Walcott, 2007). The commercial and legal stakes are high. Much effort has been devoted to finding reliable targets for PCR-based detection, often drawing on genomic sequencing and high-thoughput resequencing of diverse strains. But if PCR, ELISA, selective culturing, and other tests remain insufficiently sensitive, is this due solely to the needle-in-a-haystack sampling challenges? Are there paradigm-shifting methods available from other subdisciplines like bioterrorism prevention that are waiting to be applied to phytobacteriology?
We conclude by offering a few specific suggestions to increase exploration of new niches.
Shake Up the Review Panels
Understandably, funding agencies often enlist researchers who currently receive funding from that agency to serve on their proposal review panels. However, this practice may reinforce a narrow vision of research excellence. To increase research on underexplored niches, some panel managers have successfully broadened their portfolio by recruiting panelists from outside of their funded community, including researchers from significantly different disciplines as well as recent applicants whose unfunded proposals were regarded as highly creative or novel.
Train for Breadth
Consciously multidisciplinary training will increase the likelihood that our students and postdoctoral researchers become scientists who think broadly and are eager to work with partners who have a very different perspective or toolset. Professors can encourage this by broadening coverage in their own courses and modeling broad collaboration in their research programs. Students and postdocs can generate breadth through their course selections, their reading, meeting, and seminar choices, and through active pursuit of collaborative research.
Support Outdoor Science
Finally, we will not succeed in these underexplored niches if molecular biology lab rats do not work with colleagues who spend time in the field. Scientists with a strong laboratory orientation can benefit enormously from the biological expertise and thoughtful perspectives of field pathologists. Find the time to chat regularly with these colleagues. Moreover, the ongoing loss of extension agents and applied plant pathologists through retirements and funding cuts imperils this entire field of study. Relevant insights from natural and agricultural environments will dry up if we do not give our strongest moral and practical support to scientists with expertise in field biology.
Acknowledgments
The authors acknowledge helpful comments from Patricia McManus and support from the University of Wisconsin-Madison College of Agricultural and Life Sciences.
Literature Cited
Araújo WL, Marcon J, Maccheroni W Jr, Van Elsas JD, Van Vuurde JW, Azevedo JL (2002) Diversity of endophytic bacterial populations and their interaction with Xylella fastidiosa in citrus plants. Appl Environ Microbiol 68: 4906–4914
Bent A, Mackey D (2007) Elicitors, effectors, and R genes: the new paradigm and a lifetime supply of questions. Annu Rev Phytopathol 45: 399–436
Billing E (1987) Bacteria as Plant Pathogens, Vol 14. American Society for Microbiology, Washington, DC
Boch J, Joardar V, Gao L, Robertson TL, Lim M, Kunkel BN (2002) Identification of Pseudomonas syringae pv. tomato genes induced during infection of Arabidopsis thaliana. Mol Microbiol 44: 73–88
Brencic A, Winans SC (2005) Detection of and response to signals involved in host-microbe interactions by plant-associated bacteria. Microbiol Mol Biol Rev 69: 155–194
Brown DG, Allen C (2004) Ralstonia solanacearum genes induced during growth in tomato: an inside view of bacterial wilt. Mol Microbiol 53: 1641–1660
Campos E, Maher EA, Kelman A (1982) Relationship of pectolytic clostridia and Erwinia carotovora strains to decay of potato tubers in storage. Plant Dis 66: 543–546
Charkowski AO (2009) Decaying signals: will understanding bacterialplant communications lead to control of soft rot? Curr Opin Biotechnol 20: 178–184
Christner BC, Morris CE, Foreman CM, Cai R, Sands DC (2008) Ubiquity of biological ice nucleators in snowfall. Science 319: 1214
Craciun G, Tang YZ, Feinberg M (2006) Understanding bistability in complex enzyme-driven reaction networks. Proc Natl Acad Sci USA 103: 8697–8702
Denny T, Baek S (1991) Genetic evidence that extracellular polysaccharide is a virulence factor of Pseudomonas solanacearum. Mol Plant Microbe Interact 4: 198–206
Dulla G, Lindow SE (2008) Quorum size of Pseudomonas syringae is small and dictated by water availability on the leaf surface. Proc Natl Acad Sci USA 105: 3082–3087
Gantner S, Schmid M, Durr C, Schuhegger R, Steidle A, Hutzler P, Langebartels C, Eberl L, Hartmann A, Dazzo FB (2006) In situ quantitation of the spatial scale of calling distances and population densityindependent N-acylhomoserine lactone-mediated communication by rhizobacteria colonized on plant roots. FEMS Microbiol Ecol 56: 188–194
Gibson KE, Kobayashi H, Walker GC (2008) Molecular determinants of a symbiotic chronic infection. Annu Rev Genet 42: 413–441
Gilbert GS, Parke JL, Clayton MK, Handelsman J (1993) Effects of an introduced bacterium on bacterial communities on roots. Ecology 74: 840–854
Gilbert GS, Parke JL, Handelsman J, Clayton M (1996) Use of cluster and discriminant analyses to compare rhizosphere bacterial communities following biological perturbation. Microb Ecol 32: 123–147
Gitaitis RD, Walcott RR (2007) The epidemiology and management of seedborne bacterial diseases. Annu Rev Phytopathol 45: 114–121
Glazebrook J (2005) Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Annu Rev Phytopathol 43: 205–227
Grenier AM, Duport G, Pages S, Condemine G, Rahbe Y (2006) The phytopathogen Dickeya dadantii (Erwinia chrysanthemi 3937) is a pathogen of the pea aphid. Appl Environ Microbiol 72: 1956–1965
Hirano SS, Charkowski AO, Collmer A,Willis DK, Upper CD (1999) Role of the Hrp type III protein secretion system in growth of Pseudomonas syringae pv. syringae B728a on host plants in the field. Proc Natl Acad Sci USA 96: 9851–9856
Hirano SS,Osterag EM, Savage SA, Baker LS,Willis DK, Upper CD(1997) Contribution of the regulatory gene lemA to field fitness of Pseudomonas syringae pv. syringae. Appl Environ Microbiol 63: 4304–4312
Holt JG (1994) Bergey’s Manual of Determinative Bacteriology, Ed 9. Lippincott Williams & Wilkins, New York, pp 179–181
Kobayashi DY, Palumbo JD (2000) Bacterial endophytes and their effects on plants and uses in agriculture. In CW Bacon, JF White, eds, Microbial Endophytes. CRC Press, New York, pp 199–229
Kotoujansky A (1987) Molecular genetics of pathogenesis by soft-rot Erwinia. Annu Rev Phytopathol 25: 405–430
Leben C, Sleesman JP (1981) Bacterial pathogens: reducing seed and in vitro survival by physical treatments. Plant Dis 65: 876–878
Lindgren P, Peet P, Panapoulos N (1986) Gene cluster of Pseudomonas syringae pv. “phaseolicola” controls pathogenicity of bean plants and hypersensitivity of nonhost plants. J Bacteriol 168: 512–522
Long TA, Brady SM, Benfry PN (2008) Systems approaches to identifying gene regulatory networks in plants. Annu Rev Cell Dev Biol 24: 81–103
Lu H, Patil P, Sluys MAV, White FF, Ryan R, Dow JM, Rabinowicz P, Salzberg SL, Leach JE, Sonti R, et al (2008) Acquisition and evolution of plant pathogenesis-associated gene clusters and candidate determinants of tissue-specificity in Xanthomonas. PLoS One 3: e3828
Melotto M, Underwood W, Koczan J, Nomura K, He SY (2006) Plant stomata function in innate immunity against bacterial invasion. Cell 126: 969–980
Meng Y, Li Y, Galvani CD, Hao G, Turner JN, Burr TJ, Hoch HC (2005) Upstream migration of Xylella fastidiosa via pilus-driven twitching motility. J Bacteriol 187: 5560–5567
Mo YY, Geibel M, Bonsall RF, Gross DC (1995) Analysis of sweet cherry (Prunus avium L.) leaves for plant signal molecules that activate the syrB gene required for synthesis of the phytotoxin, syringomycin, by Pseudomonas syringae pv. syringae. Plant Physiol 107: 603–612
Monier JM, Lindow SE (2005) Spatial organization of dual-species bacterial aggregates on leaf surfaces. Appl Environ Microbiol 71: 5484–5493
Monod J, Jacob F (1961) General conclusions: teleonomic mechanisms in cellular metabolism, growth, and differentiation. Cold Spring Harb Symp Quant Biol 26: 389–401
Morris CE, Sands DC, Vinatzer BA, Glaux C, Guilbaud C, Buffie`re A, Yan S, Dominguez H, Thompson B (2008) The life history of the plant pathogen Pseudomonas syringae is linked to the water cycle. ISME J 2: 321–334
Mount MS, Lacy GH, editors (1982) Phytopathogenic Prokaryotes. Academic Press, New York
Nakaho K, Allen C (2009) A pectinase-deficient Ralstonia solanacearum strain induces reduced and delayed structural defenses in tomato xylem. J Phytopathol 157: 228–234
Nester E, Gordon MP, Kerr A, editors (2004) Agrobacterium tumefaciens: from plant pathology to biotechnology. American Phytopathological Society Press, St. Paul
Newman KL, Almeida RP, Purcell AH, Lindow SE (2003) Use of a green fluorescent strain for analysis of Xylella fastidiosa colonization of Vitis vinifera. Appl Environ Microbiol 69: 7319–7327
Niepold F, Anderson D, Mills D (1985) Cloning determinants of pathogenesis from Pseudomonas syringae pathovar syringae. Proc Natl Acad Sci USA 82: 406–410
Osbourn A, Barber CE, Daniels MJ (1987) Identification of plant-induced genes of the bacterial pathogen Xanthomonas campestris using a promoter-proble plasmid. EMBO J 6: 23–28
Payne DJ, Cramp R, Winstanley DJ, Knowles DJ (1994) Comparative activities of clavulanic acid, sulbactam, and tazobactam against clinically important beta-lactamases. Antimicrob Agents Chemother 38: 767–772
Pérombelon MCM, Gullings-Handley J, Kelman A (1979) Population dynamics of Erwinia carotovora and pectolytic Clostridium spp. in relation to decay of potatoes. Phytopathology 69: 167–173
Rainey PB, Preston GM (2000) In vivo expression technology strategies: valuable tools for biotechnology. Curr Opin Biotechnol 11: 440–444
Riesenfeld C, Schloss PD, Handelsman J (2004) Metagenomics: genomic analysis of microbial communities. Annu Rev Genet 38: 525–552
Russo N, Robinson T, Fazio G, Aldwinckle H (2008) Fire blight resistance of Budagovsky 9 apple rootstock. Plant Dis 92: 385–391
Schloss PD, Handelsman J (2008) A statistical toolbox for metagenomics: assessing functional diversity in microbial communities. BMC Bioinformatics 9: 34
Schuster ML, Coyne DP (1974) Survival mechanisms of phytopathogenic bacteria. Annu Rev Phytopathol 12: 199–221
Sepulchre JA, Reverchon S, Nasser W (2006) Modeling the onset of virulence in a pectinolytic bacterium. J Theor Biol 244: 239–257
Smith EF (1920) An Introduction to Bacterial Diseases of Plants. W.B. Saunders and Co., Philadelphia
Starr MP (1984) Landmarks in the development of phytobacteriology. Annu Rev Phytopathol 22: 169–188
Staskawicz BJ, Dahlbeck D, Keen NT (1984) Cloned avirulence gene of Pseudomonas syringae pv. glycinea determines race-specific incompatibility on Glycine max (L.) Merr. Proc Natl Acad Sci USA 81: 6024–6028
Stermitz FR, Lorenz P, Tawara JN, Zenewicz LAKL (2000) Synergy in a medicinal plant: antimicrobial action of berberine potentiated by 5’-methoxyhydnocarpin, a multidrug pump inhibitor. Proc Natl Acad Sci USA 97: 1433–1437
Upper CD, Hirano SS (1996) Predicting behavior of phyllosphere bacteria in the growth chamber from field studies. In CE Morris, C Nguyen-The, PC Nicot, eds, Aerial Plant Surface Microbiology. Plenum Press, New York, pp 277–284
Vidaver A (1981) Challenges in plant pathogenic bacteriology for the year 2000. In Vth International Conference on Plant Pathogenic Bacteria, August 16-23, 1981. Centro Internacional de Agricultura Tropical, Cali, Colombia, pp xxiii–xxviii
Walker JC (1963) The future of plant pathology. Annu Rev Phytopathol 1: 1–14
Wright CA, Beattie GA (2004) Pseudomonas syringae pv. tomato cells encounter inhibitory levels of water stress during the hypersensitive response of Arabidopsis thaliana. Proc Natl Acad Sci USA 101: 3269–3274
Yuan ZC, Haudecoeur E, Faure D, Kerr KF, Nester EW(2008) Comparative transcriptome analysis of Agrobacterium tumefaciens in response to plant signal salicylic acid, indole-3-acetic acid and gamma-amino butyric acid reveals signalling cross-talk and Agrobacterium-plant co-evolution. Cell Microbiol 10: 2339–2354
Zambryski P (1988) Basic processes underlying Agrobacterium-mediated T-DNA transfer to plant cells. Annu Rev Genet 22: 1–30
Zinniel DK, Lambrecht P, Harris NB, Feng Z, Kuczmarski D, Higley P, Ishimaru C, Arunakumari IA, Barletta RG, Vidaver AK (2002) Isolation and characterization of endophytic colonizing bacteria from agronomic crops and prairie plants. Appl Environ Microbiol 68: 2198–2208
