Dollar spot of turfgrass
Sclerotinia homoeocarpa
Warm- and cool-season turfgrasses.

Dollar spot in Zoysia grass (Courtesy A. Martinez-Espinoza)
Symptoms and signs
Symptoms
Dollar spot is a foliar disease of turfgrass. Affected grasses exhibit white to straw-colored lesions that progress downward from the leaf tip or laterally across leaf blades. A brown border usually surrounds each lesion (Figure 1). Older lesions on higher mowed grass frequently appear hourglass-shaped, being narrower in the middle than at the top or bottom. Individual leaf blades may contain many small lesions or one large lesion or the entire leaf blade can become damaged (Figure 2). Infected leaves become blighted, turning white to straw-colored as lesions expand and coalesce. Blighted leaves are formed in aggregates that appear as circular, sunken patches, measuring from < 1 to > 10 cm (< 0.5 to > 4 inches) in diameter (Figures 3-5). On golf putting greens and other closely mown areas, the patches appear as white to straw colored spots that are similar in diameter to a silver dollar, hence the name dollar spot (Figures 4-6).

Figure 1 |

Figure 2 |

Figure 3 |

Figure 4 |

Figure 5 |

Figure 6 |
Individual dollar spots may be less distinct on higher mowed turfgrasses (Figures 7-9). Dollar spots may coalesce into large straw-colored areas of blighted turf measuring 15 cm – 3 meters (6 inches to roughly 10 feet) in diameter (Figures 7-12). Dollar spot-affected turfgrass areas often become thinned of foliage and invaded by weed species. Symptoms of dollar spot, Pythium blight, and brown patch may be similar at certain stages of disease development. Usually, dollar spot is not associated with a rapid kill of turfgrass plants as are Pythium blight or Rhizoctonia brown patch. The fungi that cause dollar spot and brown patch often produce distinct lesions on infected leaves (Figure 12), but Pythium blight does not. Even though dollar spot symptoms are confined to aerial parts of turfgrass plants, the causal pathogen, Sclerotinia homoeocarpa, produces a metabolite that is toxic to bentgrass roots. The toxin causes roots to thicken, cease to elongate, and become devoid of root hairs.

Figure 7 |

Figure 8 |

Figure 9 |

Figure 10 |

Figure 11 |
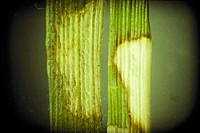
Figure 12 |
Signs
Grayish-white, cottony mycelium often forms on infected grass blades in the early morning hours when dew is present (Figures 13 and 14). Aerial mycelium produced by S. homoeocarpa is similar in appearance to mycelium produced by Pythium aphanidermatum, which causes Pythium blight, and by Rhizoctonia solani, the causal agent of brown patch. However, the latter pathogens produce foliar symptoms that are distinct from those produced by S. homoeocarpa. Foliar mycelium of S. homoeocarpa emerges from the white to straw-colored lesions, whereas abundant mycelium of Pythium and Rhizoctonia typically emerges from brown, blighted leaves. Even though the aerial mycelium of the three pathogens is similar in appearance, microscopic observation of hyphae reveals major differences. The most notable difference between S. homoeocarpa (Figure 15A), P. aphanidermatum (Figure 15B), and R. solani (Figure 15C) hyphae is the right and acute angle branching produced by Rhizoctonia hyphae. The branch hyphae appear slightly constricted at the origin of each branch, and a septum is present near a branch origin. Presence of hyphal septa can distinguish S. homoeocarpa hyphae from that of P. aphanidermatum which produces aseptate (nonseptate) hyphae (Figure 15B) except for the occasional wall formation as cells die back. Sclerotinia homoeocarpa also produces hyphae that are generally larger in diameter than hyphae of R. solani (Figure 15).

Figure 13 |

Figure 14 |
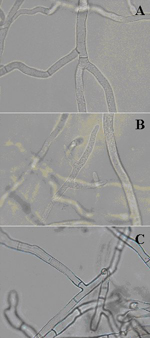
Figure 15 |
Pathogen Biology
Currently, the dollar spot pathogen is classified as Sclerotinia homoeocarpa. However, this classification is under revision, and once completed, the fungus may be reclassified as Lanzia, Moellerodiscus, or Rutstroemia. Reasons for the proposed reclassification of the fungus are as follows: 1) failure of S. homoeocarpa to form sclerotia which is a characteristic of Sclerotinia spp., 2) apothecial morphology of S. homoeocarpa differs from that of other Sclerotinia spp., 3) electrophoretic protein patterns and ribosomal DNA of S. homoeocarpa are similar to those of Lanzia, Moellerodiscus, and Rutstroemia. In culture, S. homoeocarpa produces mycelium that is white and compact. In comprehensive studies of the fungus conducted in the 1930s, isolates of S. homoeocarpa from the U.S. and Australia were sterile, while several isolates from Great Britain produced ascospores, conidia, and microconidia. S. homoeocarpa is the only member of the Sclerotinia genus that does not produce a sclerotium. Instead, the fungus produces a matrix of vegetative hyphae referred to as a stroma that can survive for long periods in grass clippings, thatch or soil.
Disease Cycle and Epidemiology
Disease Cycle
Mycelium of S. homoeocarpa can penetrate leaves directly, enter through cut leaf tips, or enter through stomata and cause infection. Hyphae colonize epidermal and mesophyll cells. The fungus secretes enzymes and toxins that result in tissue necrosis. S. homoeocarpa survives as mycelium or stromata in infected plants and is more likely to be found in thatch than in soil. When the environment is conducive for disease, mycelium grows from infected tissue and infects nearby plants. The fungus does not produce spores, so movement of mycelium or infected leaf debris by equipment, people, animals, water, or wind aids in dissemination (Figure 16).

Figure 16 |
Epidemiology
Dollar spot symptoms develop rapidly at temperatures between 15°C and 32°C (60°F and 90°F). These temperatures, combined with long periods of leaf wetness from dew, rain, or irrigation favor growth of the fungus and infection of leaf blades and sheaths. The disease is most prevalent during the spring and fall.
Low soil moisture enhances the severity of dollar spot. In one study, foliar blighting of Kentucky bluegrass (Poa pratensis) was more than doubled under conditions of low soil moisture. This probably results from drought or nutrient stress imposed on the turf, increasing host susceptibility.
Turfgrasses grown under low nitrogen fertility exhibit more dollar spot than grasses maintained at optimum fertility. Nitrogen-starved turf has been shown to be more susceptible to S. homoeocarpa infection because there is a greater amount of senescent foliage compared with turf maintained under higher nitrogen fertility. Senescent foliage provides a good food source for the fungus, and can act to increase the spread of S. homoeocarpa to healthy plant tissues.
Disease Management
Cultural practices
Turfgrass cultural practices can be used to promote an environment where infection by S. homoeocarpa is limited. Monitoring fertility is an important first step to controlling dollar spot. Turfgrasses that are maintained under low nitrogen fertility are the most susceptible to infection from S. homoeocarpa, and they are slow to recover from dollar spot injury. Light and frequent nitrogen applications are recommended for disease management and maintenance of turfgrass growth. However, over-fertilization with nitrogen may cause an increase in the overall number of dollar spot infections, and also promote other diseases such as Pythium blight and Rhizoctonia brown patch.
Drought-stressed turf is particularly susceptible to S. homoeocarpa infection. Maintaining soil moisture near field capacity (-0.033 Mpa) will limit disease severity. When irrigation is required, enough water should be provided to achieve deep soil penetration. Irrigating in the late afternoon or evening should be avoided as this prolongs overnight periods of leaf wetness. Thatch layers should be removed if they are greater than 1.25 cm (0.5 inch) in depth. Thatch can be removed by vertical mowing and topdressing with sand or soil. Controlling thatch can improve drainage, reduce drought and nutrient stress, and remove sources of Sclerotinia inoculum. Compacted soil stresses the plants and slows turfgrass growth and recovery from disease, so routine cultivation is recommended. Excessively low mowing heights also stress turfgrass and favor dollar spot. It is also recommended that adjacent trees and shrubs be pruned to promote good air movement and accelerate drying of the turfgrass canopy. The removal of morning dew by either light irrigation or poling will help dissipate leaf guttation fluids, which are nutrient-rich and provide S. homoeocarpa with a suitable growth medium. Reduced dollar spot incidence has also been observed after rolling regardless of whether dew and/or guttation fluids are present.
Chemical control
Numerous fungicides are labeled for dollar spot control, including fungicides in the following classes: benzimidazoles, demethylation inhibitors (DMIs), carboximides, dicarboximides, dithiocarbamates, succinate dehydrogenase inhibitors (SDHIs), and nitriles (Figure 17). In addition, the dinitro-aniline fluazinam controls dollar spot. Fungicides should be applied when environmental conditions are favorable for disease development. Label rates of fungicides should be applied at either a 7-10 day or 14-21 day interval. Repeated use of some chemicals, particularly benzimidazoles, dicarboximides and demethylation inhibitors, has allowed resistant populations of S. homoeocarpa to develop. Populations with resistance to multiple fungicide classes have also been reported. To limit the possibility of fungicide resistance, alternate the use of fungicides from different chemical classes (Figure 18).

Figure 17 |

Figure 18 |
Biological control
Extensive research has been conducted into the biological control of dollar spot. The disease has been suppressed in turfgrass research trials by applying composted materials, the bacterium Enterobacter cloacae or the fungus Fusarium heterosporum. Research has also been conducted on the application of nonpathogenic strains of S. homoeocarpa. These strains interact with pathogenic strains and disease is reduced. Actinovate (Streptomyces lydicus WYEC108), Armortech Sonnet (Bacillus subtilis QST713), BioJect Spot-Less (Pseudomonas aureofaciens), Companion (Bacillus subtilis GB03), Civitas (mineral oil derivative), Double nickel LC (Bacillus amyloliquefaciens D747), EcoGuard (Bacillus licheniformis SB3086), Rhapsopdy (Bacillus subtilis QST713), and Regalia (plant extract of Reynoutria sachilanensis) are biological control products that are currently registered for dollar spot control. Research has also been conducted with some success using several composted materials including turkey litter, sewage sludge, and uncomposted blends of plant and animal meals. Disease suppression has been achieved with these products in turf exhibiting light to moderate dollar spot infestations.
Resistant species and cultivars
There are some resistant species and cultivars of commonly used turfgrasses available (Figure 19). Among the cool-season grasses, perennial ryegrass (Lolium perenne) and tall fescue (Festuca arundinacea) are less susceptible to S. homoeocarpa than other commonly used species. Colonial bentgrass (Agrostis tenuis) is less susceptible than creeping bentgrass (Agrostis palustris). Annual bluegrass (Poa annua) is more susceptible than bentgrasses. Cultivars of creeping bentgrass vary in their susceptibility to S. homoeocarpa, but none is highly resistant. Among the warm-season grasses, dollar spot is particularly severe on bermudagrass (Cynodon dactylon) and seashore paspalum (Paspalum vaginatum). Consult local turfgrass extension personnel for information on resistant species and cultivars when establishing a new turfgrass area. In addition, information on the level of susceptibility of specific turfgrass species and cultivars can be obtained from the National Turfgrass Evaluation Program (www.ntep.org).

Figure 19 |
Historical Significance
Dollar spot was initially described as a disease in the 1920s when the causative agent was first thought to be a species of Rhizoctonia. The disease was initially referred to as “small brown patch” to distinguish it from “large brown patch” which is caused by R. solani. It was not until the 1930s that the causal agent was reclassified as Sclerotinia homoeocarpa. The disease is called dollar spot because spots of diseased turf are approximately the size of a silver dollar.
Dollar spot is a worldwide problem and affects almost all cultivated turfgrasses. The disease is most important in the northern areas of the U.S. on golf putting greens and fairways composed of creeping bentgrass and annual bluegrass. However, dollar spot is also damaging to other turfgrasses in home lawns, athletic fields, and other turfgrass areas. In the southeastern U.S., warm-season turfgrasses may be severely affected, but the disease occurs infrequently in the northwest and southwest U.S. In Great Britain, the disease is confined to fine-leaf fescues (Festuca spp.).
Worldwide, more money is spent on the chemical control of dollar spot than any other turfgrass disease. However, over the years, the repeated use of some fungicides has selected for fungicide-resistant populations of S. homoeocarpa. Resistance to benzimidazole and dicarboximide fungicides was reported in 1973 and 1983, respectively. Resistance to sterol demethylation inhibitor fungicides, a class commonly referred to as DMIs, was initially detected in the U.S. in 1992.
Selected References
Couch, H.B. 1995. Diseases of Turfgrasses. Krieger Publishing Company, Malabar, FL.
Detweiler, A.R., J.M. Vargas, Jr., and T.K. Danneberger. 1983. Resistance of Sclerotinia homoeocarpa to iprodione and benomyl. Plant Dis. 67: 627-630.
Endo, R.M. 1966. Control of dollar spot of turfgrass by nitrogen and its probable basis. Phytopathology 56: 877.
Giordano, P.R., T.A. Nikolai, R. Hammerschmidt, and J.M. Vargas, Jr. 2012. Timing and frequency effects of lightweight rolling on dollar spot disease in creeping bentgrass putting greens. Crop. Sci. 52:1371-1378.
Golembiewski, R.C., J.M. Vargas, Jr., A.L. Jones, and A.R. Detweiler. 1995. Detection of demethylation inhibitor (DMI) resistance in Sclerotinia homoeocarpa populations. Plant Dis. 79: 491-493.
Goodman, D.M. and L.L. Burpee. 1991. Biological control of dollar spot disease of creeping bentgrass. Phytopathology 81: 1438-1446.
Lee, J.H., G.Y. Min, G. Y. Shim, D. S. Kim, H. Sang, G. Jung, and Y. Kwak. 2015. Toothpick-aided detection of Sclerotinia homoeocarpa in the turfgrass leaf canopy, thatch, and soil in relation to dollar spot infection centers. Weed & Turfgrass Sci. 4:376-382.
Nelson, E.B. and C.M. Craft 1991. Introduction and establishment of strains of Enterobacter cloacae in golf course turf for the biological control of dollar spot. Plant Dis. 75: 510-514.
Nelson, E.B. and C.M. Craft. 1992. Suppression of dollar spot on creeping bentgrass and annual bluegrass turf with compost-amended topdressings. Plant Dis. 76: 954-958.
Smiley, R.W., P.H. Dernoeden, and B.B. Clarke. 1992. Compendium of Turfgrass Diseases 2nd ed. American Phytopathological Society, St. Paul, MN.
Smith, J.D., N. Jackson, and A.R. Woolhouse. 1989. Fungal Diseases of Amenity Turf Grasses. E. & F.N. Spon, Ltd. New York, NY.
Vargas, J.M., Jr. 1994. Management of turfgrass diseases. 2nd ed. CRC Press, Boca Raton, FL.
Vargas, J.M., Jr., R. Golembiewski, and A.R. Detweiler. 1992. Dollar spot resistance to DMI fungicides. Golf Course Management 60: 50-54.
Williams, D.W., A.J. Powell, P. Vincelli, and C.T. Dougherty. 1996. Dollar spot on bentgrass influenced by displacement of leaf surface moisture, nitrogen, and clipping removal. Crop Sci. 36: 1304-1309.
Zhou, T. and G.J. Boland. 1998. Suppression of dollar spot by hypovirulent isolates of Sclerotinia homoeocarpa. Phytopathology 88: 788-794.
